New Lymphocyte Genome Sensitivity assay can potentially identify patients with any cancer
Posted: 17 September 2015 |
Andor Komet software characterises ‘Comets’ to detect Cancer…

With cancer rates rising worldwide, and Cancer Research UK predicting that the lifetime risk of cancer will reach 50% by 2027, the need for early diagnosis is overwhelming. Now, a British research group has published details of a simple empirical test to detect any early-stage cancer, relying on Andor’s Komet software to automatically score the modified 3D ‘Comet’ assay results.
Led by Professor Diana Anderson of Bradford University and Dr Mojgan Najafzadeh of Bradford Royal Infirmary, the team has demonstrated that lymphocytes from patients suffering from melanoma, colon cancer and lung cancer are measurably more sensitive to DNA damage compared to those from healthy individuals. They also detected intermediate values from patients only suspected with melanoma, familial polyposis and chronic obstructive pulmonary disease (COPD).
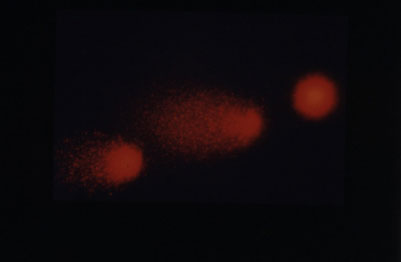
According to Professor Anderson, “The comet assay is a standard method for assessing DNA genomic damage, relying on the electrophoretic movement through agarose of supercoiled loops of DNA linked to the nuclear matrix from lysed cells. The result is structures resembling miniature comets that are observable by fluorescence microscopy, in which high genomic damage is indicated by longer optically intense tails and lower genomic damage by shorter tails.
“In our Lymphocyte Genome Sensitivity (LGS) assay, lymphocyte samples are embedded at different depths within the agar gel and irradiated with UVA in vitro. Since UVA-induced genetic damage is intensity dependent, spreading the lymphocytes 3-dimensionally within the agar means we can easily subject the cells to differing UVA intensities. Then, analysing comet tail length and fluorescence intensity determines the genomic responses to UVA.”
Automatic analysis of each comet’s percentage tail DNA and Olive Tail Moment – a measure of comet tail length multiplied by the intensity of fluorescence – for every treatment at every depth is carried out by Andor’s Komet software.
“This is vital, as percentage tail DNA is considered the most reliable comet measurement, with OTM being the most statistically significant,” says Prof. Anderson. “Since the Komet software delivered both, we could derive a linear relationship for the DNA break frequency over a wide level of DNA damage.”
According to Orla Hanrahan of Andor, “Automation is essential if the LGS assay is to be used as a screening test. Although the identification of biomarkers for specific cancers is likely to accelerate, a pre-screen to determine which patients should use these assays is required. The LGS assay developed in Bradford has sensitivities and specificities comparable to those of cancer-specific assays and, unlike the cancer-specific assays, can potentially identify patients with any cancer.”