Growth remedy: Accelerating drug performance by prioritising patient opportunities
Posted: 13 June 2016 | | No comments yet
In this article, Joaquim Nascimento discusses a three-step process to identify and prioritise initiatives to improve drug sales…

To help firms overcome tough market conditions, 20/20 Strategy outlines an approach to maximise growth for pharma brands by uncovering high-potential growth opportunities.

In recent years, it has been increasingly difficult for pharmaceutical brands to sustain high growth rates. The environment has been particularly difficult, with reduced healthcare spending, tougher access & price negotiations, or increased generic penetration eroding value of the pharmaceuticals drug market. For example, sales of prescription drugs in 2014 declined by 2% in the top 5 European markets (source: IMS Drug Monitor).
Given these challenges, pharma companies need to revise the commercialisation strategy of new and existing brands to identify additional pockets of growth, and prioritise initiatives with the best return on investment.
However, with a large number of stakeholders and with a wide range of potential initiatives, making decisions on where to allocate resources is challenging. Should brands focus on ensuring the drug is included in local or hospital formularies? Or should brands develop campaigns to improve awareness of available treatments? Or win share of physician 1st line treatment? These are some of the questions that need to be clarified to drive growth. Pin-pointing where to focus, and clarifying how the brand priorities change over time will unlock additional growth.
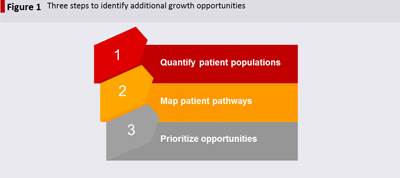
In our work with leading pharmaceutical companies across therapeutic categories and markets, we have developed a three-step process to identify and prioritise initiatives to improve drug sales: Quantify patient populations; Map patient pathways; Prioritise opportunities.

Quantify patient populations
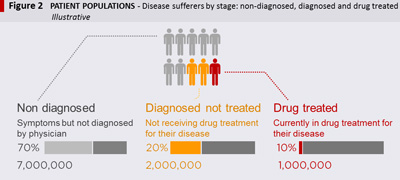
To identify the most valuable initiatives, pharma companies should adopt a patient-centred view. There is a limited pool of patients to source into each drug, so knowing how many patients are in the market and how many reside in each step of the treatment paradigm is imperative. It is not enough to know how many prescriptions are filled every month, as many market dynamics will remain uncovered – such as diagnosed patients without drug treatment, patients on drug-holidays or patients on combination therapy. In the example of a chronic disease with low treatment rates, it will be important to quantify how many patients have the disease; how many have been diagnosed and how many are currently on drug treatment (see Figure 2 – Patient Populations). Quantifying patient populations helps to clarify the potential scope of the opportunity.
Map patient pathways
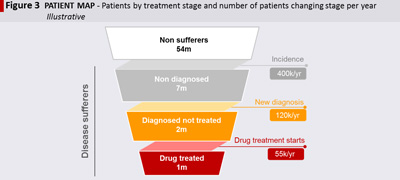
Knowing how many patients reside in each treatment stage is only the starting point. To understand where to act is important to quantify how many patients are moving through the different stages until they reach the brand, e.g.: how many new patients have the disease every year (incidence); how many are newly diagnosed; how many initiate treatment (see figure 3 – Patient map).
In this example there might be seven million sufferers who are undiagnosed and not being treated. This is a sizeable population of potential patients, however if there are significant hurdles in diagnosing this population (e.g.: patients do not recognise disease symptoms, or social stigma prevents them from discussing it with their physician), the rate of diagnosis will be low, and it will be very hard to source them. So, in order to determine which opportunities to focus on, it is critical to also quantify how many patients are moving through the different stages every year.
For most therapy classes it will be important to break-down the treated population in the Patient Map in more detail, in order to identify where individual brands source patients from. For example, the treated population could be further split into your brand vs. competitors; and/or 1st line treatment vs. later lines of treatment; or generics vs. branded. The purpose is to develop a Patient Map structure which depicts the key market dynamics and physician prescribing patterns.
At each treatment transition point (e.g., new diagnosis, drug treatment starts), it is important to understand the influencing factors, hurdles and triggers which may impact the rate at which patients move through to the next stage. This will help companies to determine the most appropriate actions to focus on. For example, drug treatment start rate and the choice of drug prescribed is influenced by a range of factors including the severity of the patient condition, physician knowledge and drug preference, payer restrictions, or patient preference.
Prioritise opportunities
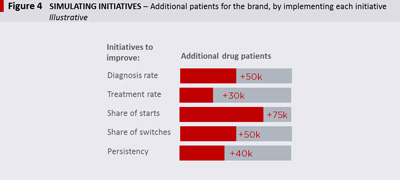
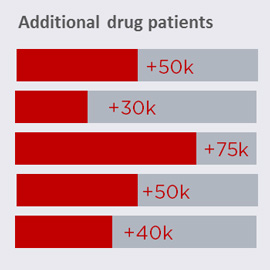
Improving performance at the key transition points in the Patient Pathway Map will ultimately impact brand sales (e.g.: the higher the diagnosis rate, the more patients will eventually start drug treatment). Once the Patient Pathway Map is populated with data on how patients move through the system, a simulation tool is used to evaluate where most value is to be won. This is done by quantifying the incremental patients and drug sales resulting from improvements at each point in the pathway. The example in Figure 4 illustrates the additional patients on a specific brand, resulting from improving performance in different areas. In this example, focusing on improving the brand’s share of starts offers the most attractive opportunity to focus on.
The simulation tool can also be used to explore what-if scenarios and test the impact of initiatives on brand performance under a range of plausible market scenarios, such as the impact of a new entrant into the category.
Knowing which opportunities to pursue provides several benefits to pharma companies:
- Outlines a road-map to growth by identifying the strategic priorities;
- Quantifies growth potential and informs forecasting;
- Gets buy-in from internal stakeholders and aligns team into a single direction;
- Provides direction for further research to inform how to communicate with stakeholders and activate the opportunity.
Based on our experience, by applying this rigorous data-driven approach across different therapeutic categories, pharma companies have been able to uncover previously unrecognised opportunities, and devise high-powered initiatives to capture growth, even in mature markets.
About the author Joaquim Nascimento is a Partner at 20/20 Strategy, specialised in developing corporate and brand strategy for some of world’s leading brands. He helps companies identify growth opportunities in the Pharmaceutical, FMCG, Technology and Education industries across Europe, Asia and America. |