How to get the best out of your high pressure homogeniser
Posted: 5 September 2016 | | No comments yet
Fluid processing requirements are becoming ever more stringent, and so high pressure homogenisation is increasingly surfacing as a solution to meeting these needs…


Fluid processing requirements are becoming ever more stringent, and so high pressure homogenisation is increasingly surfacing as a solution to meeting these needs.


It is widely understood that almost every type of sample will have its own unique optimum processing parameters, due to differing particle sizes, densities, rigidity and so on. Therefore, it is of great importance ideal parameters are identified and applied during homogenisation.
In the case of Avestin’s range of high pressure homogenisers, temperature and pressure parameters are extremely customisable, allowing for a high degree of operator flexibility, in order to satisfy sample-specific processing needs.
This article aims to highlight factors users should consider, when aiming to optimally homogenise material, whilst addressing the wide range of available applications in the field of high-pressure homogenisation.
The three main parameters to consider are:
- Pressure
- Temperature
- Number of Passes
Pressure
Material presented to a homogeniser may vary in terms of particle size, density, shape, and overall pH – facets which can create unique temperature and pressure sensitivities that should be clearly understood, prior to processing. High-pressure followed by rapid pressure decrease can allow cell lysis via forces such as liquid shear, which has many uses, including DNA purification and protein recovery. It is important that the user identifies the homogenising pressure that will achieve lysis of those particular cells, as pressures that are too low may prove ineffective.
Sample types require differing homogenising pressures for cell lysis. As demonstrated in Figure 1 below, some commonly studied bacteria require varied pressures for lysis.
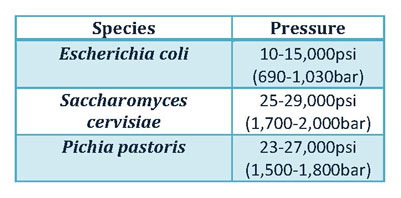

Figure 1: Commonly processed microbial organisms and their recommended homogenising pressures.
Based on the above, it is vital to identify the unique cell-lysing pressure for that particular sample. Avestin’s range of high-pressure homogenisers allows the user to adjust the homogeniser controls to the correct pressure and thus achieve desired results, with a minimal temperature increase; an occurrence that may be significant when handling heat sensitive.
Pressure and temperature are directly affected by each other. As a general rule, a 1000psi (69bar) increase in pressure results in a 1°C increase in temperature, which could potentially impact homogenisation results negatively – it is therefore important that pressure is controlled.
From bench-top to full-scale production, Avestin offer a range of units, designed with such adjustable control of pressure in mind, and thus temperature. For example, the Avestin C5 (Figure 2, right) is a lab-scale model capable of a user-defined pressure range between 500-30,000psi.


Figure 2: The Avestin C5
Of course, should clients demand higher flow-rates; Avestin’s larger scale homogenisers can easily meet these needs, with up to 1,000 litres/hour systems, again, without compromising pressure control.
Temperature
Tougher samples generally require higher pressures for processing. In the case of DNA or protein isolation, it is crucial that the optimal homogenising pressure for a particular sample does not induce a temperature high enough to damage sensitive intracellular products.
Water-in-oil emulsion stability too, can be significantly altered by temperature, due to the ease at which some physical properties of oil, water, interfacial films and surfactant solubility are impacted. An increase in temperature leads to destabilisation of crude oil/water interfacial films 1. It could also change the interfacial film build-up rate, as well as film compressibility 2.
Temperature increase may therefore induce an increase in oil viscosity and thus emulsion viscosity, so it can directly and significantly alter the quality of the end product, given optimal viscosity of an emulsion is always crucial. Avestin machines can offer that precise temperature control via compatibility with cooling coils, as well as sanitary heat exchangers.
Cooling coils (Figure 3, below) have proved an effective means of heat reduction when certain materials do not demand stringent control below 10°C, in some cases preferred to the sanitary version, as the more commercially viable choice. However, where strict temperature control is necessity, especially at lower increments ~<10°C, the sanitary heat exchanger (Figure 3, below) is recommended, due to the rigorous control associated – please note the sanitary heat exchanger must be attached to a source of cooled water i.e. chiller or re-circulator to function.


Figure 3: Cooling coil (left), sanitary heat exchanger (right)
Both are effective, and so the choice of temperature control method is dependent on the precision of the process requirement, as well commercial aspects.
Number of Passes
It should be noted that some products require high homogenising pressures for processing; however these pressures can damage sensitive products. For example, proteins may be denatured.
A solution to this problem lies in lowering the pressure and increasing the number of passes through the machine. Multi-pass, high-pressure homogenisation has been shown to induce higher microbial inactivation than single-pass high-pressure homogenisation ofL. delbrueckii 3, which may be a preference to some.
In cellular disruption, increasing the number of passes while lowering pressure can actually enable the maximum yields with active and viable products in most cases. This is has also been demonstrated with other applications such as emulsions, liposomes and dispersions, where increasing the number of passes, within reason, might improve product stability.
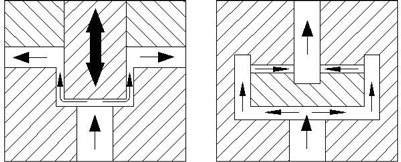

Figure 4: Avestin’s dynamic valve (left); Static valve (right).
Working in such a manner can have its downside, because, in the past, many homogenisers containing “static” homogenising valves became prone to blockage when running viscous material. In addition, processing at such high pressures leads to valve erosion, which, as well as blockage, will cost the user both time and money in terms of servicing and maintenance.
Avestin identified this issue, so developed a unique, “dynamic” self-regulating valve to counteract this occurrence (Figure 4, above). The dynamic valve maintains itself once the desired homogenising pressure is achieved. Should the valve block, the valve opens to allow the blockage to pass with little or no interruption to the process. It is this ease-of-use approach by Avestin that has made their range so popular in R&D and production environments.
In summary, the identification of the optimal parameters when processing samples via high pressure homogenisation, can allow both ease of use and cost-effectiveness.
References
1: Jones, T.J., Neustadter, E.L. and Whittingham, K.P. (1978). Water-In-Crude Oil Emulsion Stability And Emulsion Destabilization By Chemical Demulsifiers. Petroleum Society of Canada. 17 (2)
2: Quintero, C.G., Noik, C., Dalmazzone, C. and Grossiord, J.L. (2009). Formation Kinetics and Viscoelastic Properties.Oil & Gas Science and Technology. 64 (5), 607-616.
3: Franchi, M.A., Tribst, A.A.L. and Cristianini, M. (2013). High-pressure homogenization: a non-thermal process applied for inactivation of spoilage microorganisms in beer. Journal of The Institute of Brewing. 119 (4), 237-241.
Figure 1, Figure 2, Figure 3 and Figure 4 were acquired from Biopharma Process Systems Ltd.




