Laboratorios Leti S.A.V. uses Ci-DMS from CI Precision
Posted: 6 October 2016 | | No comments yet
Venezuela’s largest pharmaceutical manufacturer, Laboratorios Leti S.A.V. is using the Ci-DMS Weigh and Dispense module from CI Precision to dispense both raw and packing materials for their processing plant…
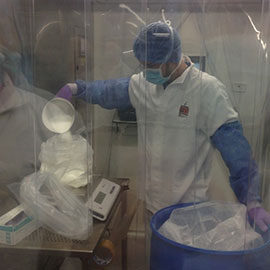

Venezuela’s largest pharmaceutical manufacturer, Laboratorios Leti S.A.V. is using the Ci-DMS Weigh and Dispense module from CI Precision to dispense both raw and packing materials for their processing plant.
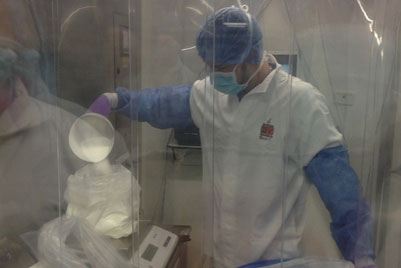

The number of dispensing booths has risen from 4 to 12 across 4 production areas, with Ci-DMS managing supply from warehouse to plant via process orders received from Leti’s SAP ERP system.
Initially implemented in 2009, the company saw Ci-DMS as offering the best solution for eliminating errors occurring in paper-based processes; improving inventory control and reducing costs; creating more efficient process workflows; and implementing compliance protocols.
With a proven history of building long-term partnerships that provide ongoing value to customers, CI Precision has worked with Laboratorios Leti to develop a number of web service interfaces for their ERP System (SAP ECC 6.0) as well as meet the demands of the business in increasing the number of dispensing booths.
Today, the company uses Ci-DMS to book all dispensing tasks and movements in SAP including receiving process orders, retrieving material QC status, dispensing materials according to GMP procedures and returning material consumption data. During weigh and dispense operations, operators follow simple on-screen instructions based on standard procedures, and electronic signatures can be invoked wherever required to control dispensing activity.
Operations Director, Cesar López Gualdrón, explains that according to statistics, before Ci-DMS implementation, 1 in every 1000 batches incurred errors such as addition of incorrect materials, errors in weight or use of incorrect lots. This represented a cost to the business in excess of $150,000 per year. Since Ci-DMS was implemented, operator errors have been avoided, saving money and production time and providing a rapid payback with a ROI of less than two years.
Henry Alarcon, IT Director, added
“We would recommend Ci-DMS as an extended manufacturing system that enables you to control processes using detailed step-by-step instructions and keep an electronic record of each event. It connects with SAP using state-of-the-art web service technology. Ci-DMS is a validated system that saves time and money and it meets the needs of our company.”
“CI Precision maintains close contact with Leti staff to resolve any support issues on time and to our satisfaction through remote connection to our SAP environment. The Ci-DMS user manuals and training are good, ensuring excellent knowledge transfer to operators and technical staff.”



