All Drug Markets articles – Page 12
-
 News
NewsFDA approval could shape liver disease treatment landscape
A novel liver therapy authorised under US Food and Drug Administration (FDA) Accelerated Approval, is a potential game-changer in the NASH treatment space.
-
 News
NewsManufacturer retracts acquisition deal due to “data integrity issues”
According to Olympus Corporation, concerns about the non-vascular metallic stents were found post-closing following the agreed acquisition of Taewoong Medical Co., Ltd.
-
 Article
ArticleThe future of targeted alpha therapy development and manufacture
Following the first industrial-scale facility for targeted alpha therapy (TAT) production in Europe beginning construction, in this exclusive Q&A, Julien Dodet, CEO of Orano Med, elaborates on the key trends in radioligand therapeutics and why TATs hold significant advantages as cancer treatments.
-
 News
NewsSandoz granted novel biosimilars approval
The approval authorises the interchangeability of denosumab biosimilars in the US to treat primary and secondary bone loss.
-
 News
NewsMedical device manufacturers must remain “vigilant” to ensure data integrity of premarket submissions
With the integrity of a greater number of third-party-generated data called into question, the US Food and Drug Administration (FDA) is unable to rely on the data to grant marketing authorisation, the agency asserts.
-
 News
NewsNew antibody drug formulation wins novel MHRA approval
A new formulation of an antibody drug has been authorised via a process from the Medicines and Healthcare products Regulatory Agency (MHRA) that enables “considerably shorter” approval timelines.
-
 News
NewsNICE recommends first licenced treatment for AL amyloidosis
Having been available in Scotland and Northern Ireland since 2022, NICE’s positive opinion of the combination treatment means it is now accessible on the NHS.
-
 Article
ArticleRare disease spotlight: first EU-approved treatment for Friedreich’s ataxia
In this exclusive article, Biogen’s Vice President and Head of the Neuromuscular Development Unit, Dr Toby Ferguson, discusses the current rare disease landscape and recent developments in treating Friedreich’s ataxia (FA).
-
 Article
ArticleMaking ATMPs a reality for rare disease patients
Advanced therapy medicinal products (ATMPs) will be among the first to undergo joint clinical assessments at EU level in 2025 and these will form the basis for national value assessments and pricing negotiations. Here, Paolo Morgese from the Alliance for Regenerative Medicine discusses how methodologies for joint clinical assessments (JCAs) ...
-
 News
NewsAmgen opens its most advanced manufacturing facility to date
The biomanufacturing facility has been designed to help Amgen meet its target of carbon neutrality in all its operations by 2027, according to the company.
-
 News
NewsCHMP meeting highlights: February 2024
Ten new medicines, including a new oligonucleotide therapy for ALS, were recommended for approval at the Committee for Medicinal Products for Human Use (CHMP)’s recent meeting.
-
 News
NewsInnovation to drive advanced drug delivery market
The advanced drug delivery market is anticipated witness a compound annual growth rate (CAGR) of 4.6 percent by 2033, according to a report.
-
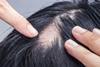 News
NewsNICE recommends first medicine for severe alopecia areata
Clinical studies of Pfizer’s oral medicine for alopecia areata have demonstrated response rates continued to improve for up to two years.
-
 News
NewsUK manufacturing output expected to improve despite challenges
Output volumes for UK manufacturing fell in the three months to February 2024, yet it is expected to rise slightly in the next three months, research reports.
-
 News
NewsSingle-use technology propelling upstream bioprocessing market expansion
A driving factor behind the expansion of the upstream bioprocessing market is the need for process improvement in the biopharma industry, research highlights.
-
 News
NewsWHO initiative to enhance biomanufacturing in LMICs
A new World Health Organization (WHO)-driven initiative aims to enhance bioproduction capabilities for biological products in low- and middle-income countries (LMICs).
-
 News
NewsPharmaceutical suspension market to witness accelerated growth
The global pharmaceutical suspensions market is set to value $83.8 billion by 2032, due to the systems having key benefits such as supporting patient compliance.
-
 News
NewsVertex secures European approval for CRISPR cell therapy
Now conditionally approved in Europe for sickle cell disease and transfusion-dependent beta thalassemia, the CRISPR therapy offers eligible patients a functional cure.
-
 News
NewsUS legislation to benefit small molecule drug innovation
Passing of two key legislations in the US is set to support innovation for small molecule drugs and the biotech sector, Biotechnology Innovation Organization (BIO) asserts.
-
 News
NewsBiogen refocuses its Alzheimer's strategy
Part of Biogen’s prioritisation of its portfolio includes focusing on the advancement of its leading anti-amyloid beta Alzheimer’s treatment, LEQEMBI® (lecanemab-irmb).



