Whitepapers
Latest Whitepapers
- Whitepaper
A software‑supported, AI‑assisted workflow for optimising LC gradient methods
Shimadzu introduces a novel automated artificial intelligence-driven HPLC method.
- Whitepaper
The critical role of CCI in drug development and manufacturing
This whitepaper provides a detailed examination of Lonza’s holistic, data-driven approach to CCI, developed in collaboration with industry experts and applied across our global network for parenteral combination products.
- Whitepaper
TCS revolutionize clinical trial monitoring and data analysis with AR/VR-driven immersive analytics
AR/VR solution for visualizing multi-dimensional data in 3D. Explore sites on a 3D globe to detect trends and anomalies with ease.
- Whitepaper
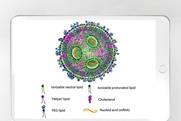
Strategies for endotoxin testing of RNA-LNP
Overcome matrix interferences in RNA–LNP endotoxin testing with LAL-based methods & recombinant factor C assays. Learn more in this whitepaper.
AI - a game changer in unifying clinical data
The use of AI technology in unifying clinical data across systems for making clinical trials more effective, safer and more efficient.
Accurate and Reproducible Quantitation of xC/UV/MS Data from Any Instrument
Learn how Medtronic processes and quantifies their xC/UV/MS data using the quantitation workflow in MS Workbook Suite.
Improving Method Transfer Outcomes
A collaboration with Pfizer to create a tool to ensuring seamless method transfer between labs.
Deepening Digitalization in Drug Discovery at PTC Therapeutics
PTC Therapeutics accelerates drug discovery process by integrating physicochemical property predictions into their Dotmatics ELN.
Accelerating Process Chemistry
Process chemists can increase their efficiency by accessing process, analytical, and chemical data from across their organization using Luminata.
More Whitepapers
Standardization of Analytical Data: Best Practices
Learn about available formats for analytical data standardization, limitations, and how to ensure data is usable by lab scientists and AI/ML.
NSF Launches ‘Compliance Unlocked’ Podcast Series
NSF invites pharmaceutical professionals to tune in to its new groundbreaking podcast series “Compliance Unlocked” and join the conversation on critical industry issues.
Cell and Gene Therapy Containment
Overcoming Challenges in Cell and Gene Therapy Containment.
NSF’s quality management maturity (QMM) assessment model
NSF’s quality management maturity (QMM) assessment model: future-proofing quality and supporting supply chain robustness
Unlock exclusive industry insights
- Bookmark articles and resources to access anytime
- Enjoy free access to industry leading resources, webinars and insights
- Stay informed with the latest news and breakthroughs in drug discovery
- Receive updates and recommendations tailored to your research interests
Understanding and Applying Updated FDA Guidance
Understanding and Applying the Updated FDA Guidance: Inspection of Injectable Products for Visible Particulates.
Regulatory Authority Inspection Comparison
A guide on what to expect during a regulatory inspection and the differences in the approach taken by three of the main regulatory authorities and PIC/S.
Application note: The need for rapid methods in the compounding pharmaceutical industry
This application note explores rapid microbial methods and balancing patient care, quality products, and safe shelf lives for unique formulations.
Selecting Fill/Finish Manufacturing Technologies
With a growing pipeline of biologics, increase in vaccine production, and small batch sizes targeting small patient populations, there’s increasing demand for advanced manufacturing tech.
Whitepaper: What to expect from a regulatory inspection, an NSF expert guide
Regulatory inspections can be challenging, but proper preparation turns them into opportunities for improvement and compliance demonstration.
Whitepaper: Risk-Based Approach to Development - Injectable Combination Product
Drug-device combination products offer advantages for patients but are complex and challenging to develop. Establish a holistic approach to DDCP development and assess and manage risk throughout the process.
Whitepaper: Optimise organisational culture & quality maturity
Whitepaper - Optimising organisational culture and quality maturity is crucial in promoting consistent, reliable business processes and to minimise supply disruptions.
Delivering market success for concept to patient
Whitepaper - Delivering Market Success for Pharma Partners from Concept to Patient
A data-driven approach to Container Closure Integrity
Selecting container closure components with confidence:A data-driven approach to Container Closure Integrity.
Application note: Single temperature analysis for environmental monitoring samples
This application note explores how to determine the feasibility and the impact of the single temperature for environmental monitoring microorganisms.
eBook: From Vial to Prefilled Syringe
Simplify your move from a vial to a prefilled syringe – lifecycle planning for containment and delivery of biologics drug products
eBook: Cut through complexity in drug development ebook
How you can cut through complexity, gain speed and get your drug to the patient as soon as possible.
eBook: Successfully navigating complex and evolving regulatory environment for combination products
With complex processes and stringent regulatory requirements, the path to developing a combination product can be uncertain and challenging
Science of scale for spray-dried intermediates
Speed to market is a critical aspect of developing new pharmaceutical products, and scale-up – or scale-down – can play a key part in that process.
Whitepaper: USP <645> Water Conductivity Regulations
This whitepaper looks at benefits of measuring conductivity on-line, and calibration and temperature compensation criteria for USP compliance.
Synthesis and Route Design to Tackle API Complexity
These longer synthetic pathways present challenges for process chemists hoping to achieve an efficient API manufacturing process.
Application note: Highly efficient method development using LC-MS for automated peak tracking
Peak tracking in liquid chromatography (LC) method development can be challenging due to retention variations and co-elution. LabSolutions MD software with LCMS-2050 SQ-MS simplifies peak identification and automates method and sequence creation, enabling efficient development of reliable separation methods.
Advertorial: Next-generation microbial testing of pharmaceuticals
Specialised microbial testing is key in drug development to mitigate contamination risks, which threaten drug efficacy and patient safety. Bruker's MALDI Biotyper® at MPL, Austria, ensures accurate and robust testing, in a high-throughput workflow, delivering reproducible results.
Application note: Automated nucleic acid amplification assay for mycoplasma detection in cell and gene therapy products
Ambili Menon, Pharma Quality Control, bioMérieux Inc. addresses the task of microbiological testing for advanced therapeutic medicinal products.
Application note: Validating recombinant cascade reagents in 3 simple steps
Endotoxin testing continues to evolve alongside the industry’s need for more sustainable methods that reduce ecological impact. Nicola Reid, Associate Director of Endotoxin Products, Charles River Labs, reflects on these developments which are driven by the 3Rs (replacement, reduction and refinement).
One HEK of a viral vector to develop
Viral vector manufacturing presents many challenges related to HEK cell line development, fluid transfer and product viability in the cold chain.
Case study: Data-driven water system maintenance
This case study looks at how an online microbial analyzer provides real-time bioburden monitoring for rapid response to adverse water system events.
Case Study: Franz Ziel’s utilisation of Enzyme Indicators using Fractional Kill Time method
An EJPPS published study on Enzymatic Indicators in Vaporized Hydrogen Peroxide Decontamination Cycles: Application-related Research focusing on Fractional Kill Time (FKT) and Reverse Fractional Kill Time (RFKT) Studies
Whitepaper: Molecular contamination control - odours, gases and toxics
Identify molecular contamination concerns in the pharmaceutical workspace and determine the right filtration solution for dealing with these problems.