All Drug Markets articles – Page 4
-
 Article
ArticleWhat will shape the Pharma landscape in Q3 2025?
Upcoming drug approvals and clinical progress will be pivotal during the third quarter of the year, says GlobalData.
-
 News
NewsMHRA approves needle-free allergy drug alternative
The MHRA's approval provides eligible patients with a novel delivery method that is convenient and non-invasive.
-
News
Boehringer partnership to advance biologic for rare skin condition
The new partnership between Boehringer Ingelheim and LEO Pharma aims to expand treatment access for more patients with generalised pustular psoriasis.
-
 News
NewsUK Government publishes Life Sciences Sector Plan
While a focus on accelerating access for innovative medicines has been welcomed, industry representatives caution that current system infrastructure could hinder the plans proposed by the UK Government.
-
 News
NewsNICE recommends innovative cystic fibrosis therapy
Recommendation of the once-daily triple therapy increases the number of eligible cystic fibrosis patients who can access treatments in the UK.
-
 News
NewsUpdated licensing agreement marks “major milestone” in global HIV response
The expanded agreement between ViiV Healthcare and Medicines Patent Pool (MPP) provides a more patient-centric treatment approach for HIV patients worldwide.
-
 News
NewsTechnology advancement to drive lab automation market through next decade
While high costs are hindering growth in the lab automation market, research highlights that new innovations like AI offer potential to expand this rapidly evolving industry.
-
 News
NewsMSD acquisition to accelerate first-in-class COPD drug
The $10 billion acquisition involves advancement of a novel inhaled therapy which has potential to redefine standard of care in respiratory disease.
-
 News
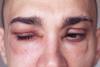
NewsFDA approves first-of-a-kind oral therapy for rare disease
The US drug agency’s authorisation provides hereditary angioedema (HAE) patients with the first new on-demand treatment in over ten years.
-
 News
NewsNext drug patent cliff to challenge Big Pharma strategy
While the pharmaceutical market is expected to witness strong pricing competition, there is also significant opportunity for biotech companies, research says.
-
 News
NewsSandoz breaks ground on new European biosimilar plant
The new investment will help Sandoz to expand its European biosimilar manufacturing capacity and ensure reliable and sustainable supply in the region.
-
 Article
ArticleAlliance for impact - advancing CGT development in Europe
Cell therapy expert Lindsay Davies, PhD, FHEA, discusses the key considerations for moving the needle to broader adoption of CGTs in Europe.
-
 News
NewsGilead partners to advance novel oral oncology drug
The deal between Gilead Sciences and Kymera Therapeutics could lead to development of safer and more effective cancer treatments.
-
 Report
ReportStreamlining European expansion for cell and gene therapies
Tom Smith, Strategic Director, Cell and Gene Therapies, Uniphar, discusses the essential elements biotechs need for commercial success in Europe’s unique market when developing cell and gene therapies (CGTs).
-
 News
NewsLife sciences a “priority sector” in UK Industrial Strategy
The new UK strategy represents a milestone for enhancing international competitiveness and puts into motion the upcoming Life Sciences Sector Plan.
-
 News
NewsRegulatory shift signals potential change for future CGT sector
The cell and gene therapy (CGT) sector must maintain its scientific rigor despite recent changes to US policy and market conditions, says key industry body.
-
 News
NewsNICE concludes decision for two Alzheimer’s treatments
The committee’s final draft guidance cited high-cost as a key reason for its negative opinion of the Alzheimer’s drugs, developed by Biogen and Eisai.
-
 Article
ArticleNew biosimilar pathways – key takeaways from the EMA’s draft reflection paper
Gain insight on the European Medicines Agency (EMA)’s latest draft reflection paper and its scientific rationale, regulatory implications and potential impact on biosimilar drug development and manufacturing in the EU.
-
 News
NewsMHRA Class 2 medicine recalls – May/June 2025
The UK medicine agency's most recent recalls of this class have been due to packaging and potential contamination issues.
-
 News
NewsMHRA issues first UK guidance on phage therapies
MHRA’s publication is intended to support European developers of phage-based medicinal products to effectively navigate related regulations.



