All Emblaveo articles
-
 News
NewsCHMP meeting highlights: March 2024
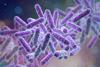
At the Committee for Medicinal Products for Human Use (CHMP)’s March meeting, twelve medicines received positive opinions for marketing authorisations, including a new antibiotic and a novel oral medicine for a rare blood disorder.
-
 News
NewsPositive CHMP recommendation for antibiotic combination
EMA has recommended granting a marketing authorisation for Emblaveo for treatment of infections caused by multidrug-resistant bacteria.



