NICE gives green light to two eye treatments for treating diabetic macular oedema
Posted: 22 July 2015 |
NICE has recommended aflibercept solution for injection and dexamethasone implant as treatment options for some people with diabetic macular oedema…
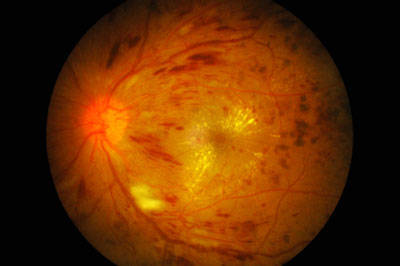

The UK National Institute for Care and Health Excellence (NICE) has recommended Bayer’s aflibercept (Eylea) solution for injection and Allergan’s dexamethasone (Ozurdex) intravitreal implant as treatment options for some people with diabetic macular oedema, in two separate pieces of final guidance published today.

Diabetic macular oedema is a common complication associated with diabetic retinopathy, and is the most common cause of visual impairment in people with diabetes. It develops when blood vessels in the eye are damaged by a continuously high blood sugar level, causing plasma to leak into the eye. Abnormal new blood vessels also form at the back of the eye (known as the retina). This causes a build-up of excess fluid (oedema) in the macular, which is the central part of the retina. The oedema impairs light perception, causing blurred vision. If it is not treated effectively, it can lead to irreversible vision loss.
Aflibercept is a vascular endothelial growth factor (VEGF) inhibitor. It prevents abnormal new blood vessels from forming under the retina. It also helps treat swelling in the retina caused by oedema.
Diabetic macular oedema affects around 189,000 people in the UK
Dexamethasone is a corticosteroid that works by suppressing inflammation and preventing oedema forming in the eye. It reduces plasma leakage from blood vessels and inhibits the release of inflammatory compounds that cause damage to the eye.
The dexamethasone intravitreal implant is injected into the eye, with each implant delivering around 700 micrograms of dexamethasone to the back of the eye over a period of 6 months or more. The implant remains in the vitreous for up to 270 days before fully dissolving. After initial treatment, re-treatment can be performed after approximately 6 months if the patient experiences decreased vision with or without an increase in retinal thickness with recurrent or worsening diabetic macular oedema.
Professor Carole Longson, Health Technology Evaluation Centre Director at NICE said, “NICE is pleased to approve both dexamethasone intravitreal implant and aflibercept as treatment options for some people with diabetic macular oedema in final guidance. The condition affects around 189,000 people with diabetes in the UK, and can have a substantial negative impact on quality of life and daily activities.
“Whilst these recommendations are for people with diabetic macular oedema needing different treatments at different stages of the disease, these decisions will be welcome news to both patients and healthcare professionals alike.”
Related organisations
National Institute for Health and Clinical Excellence (NICE)



