Tom Smith, Strategic Director, Cell and Gene Therapies, Uniphar, discusses the essential elements biotechs need for commercial success in Europe’s unique market when developing cell and gene therapies (CGTs).
This ‘classical build-out’ model was designed for high-volume launches and broad portfolios, so is unsuitable for biotechs that may have only one or a small number of assets. Cell and gene therapies differ in that they typically target small, geographically dispersed populations and require specialist treatment and engagement.
“Cell and gene therapy companies need speed, agility, and the ability to align strategy with existing infrastructure to deliver to patients.”
Investors expect speed and capital efficiency, so it is wise to partner early and ‘lease’ the infrastructure required to launch. Uniphar, for example, offers an end-to-end European commercial platform: from Enterprise Resource Planning (ERP) systems to Quality Management Systems (QMS), distribution licenses (WDA/MIA), and local access to over 1,800 hospitals.
Uniphar is uniquely positioned to support clients at every stage of the journey, enabling them to act with full operational capability, without having to hire country-by-country teams. Biotechs can therefore not only align scientific ambition with commercial feasibility, but also gain quicker market entry and at a fraction of the cost and time.
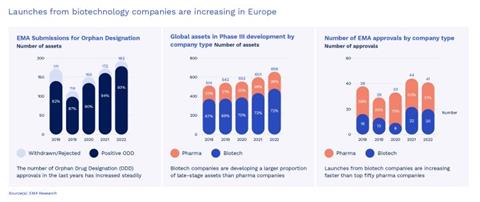
Uniphar New Opportunities Whitepaper Figure 2 ALT
This model supports two pathways: companies can either operate indefinitely using partner infrastructure or use it as a springboard while building their own. In parallel, Europe’s evolving regulatory landscape is introducing efficiencies. The 2025 implementation of the EU Joint Clinical Assessment (JCA) requires a single, pan-European clinical dossier at the time of European Medicines Agency (EMA) filing. While this centralises scientific review, pricing and reimbursement remain firmly in national hands. It is therefore vital to engage both centrally and locally, requiring deep knowledge of country-specific payer expectations and timelines.
Effective market access in Europe depends on combining central coordination with in-country execution. For example, Uniphar delivers both: a centralised team that manages pricing and reimbursement strategy across markets, and a network of local experts who have to knowhow and relationships to handle submissions in the right language, format, and context. These in-country relationships with payers and regulators are essential to navigating complex HTA processes and avoiding delays.

Another critical success factor is real-world evidence (RWE). European payers increasingly require post-launch data to validate long-term value. Uniphar supports clients in setting up registries, managing expanded access programmes, and gathering long-term outcomes, activities that are critical for both sustained reimbursement and expansion.
For today’s advanced therapy developers, speed and flexibility can mean the difference between making it to market or being left behind. Europe isn’t a barrier; with the right partner, it provides numerous opportunities. By rethinking expansion and engaging the right partner, cell and gene companies can unlock this potential quickly, affordably, and with confidence.
For further information, visit: