IMA Active explores how interconnected, human-centric systems that implement advanced automation and usability can enhance operational efficiency and sustainability in tablet manufacturing.

CDC with Domina 500 (Credit: IMA Active)
Automation in life sciences is becoming increasingly intelligent, interconnected and adaptable. However, innovation must never compromise usability.
This article outlines the principles guiding the automation development of DOMINA and SOLIDA tablet presses, emphasising human-centric interfaces, built-in data integrity, and cybersecurity aligned with Annex 11, 21 CFR Part 11 and IEC 62443. It details how a quality by design (QbD) approach, supported by process analytical technology (PAT), facilitates ongoing process verification, real-time release testing and inline quality control.
The importance of flexibility and scalability in modern production methods, including batch, semi-batch and continuous manufacturing is also discussed. Chiefly, how orchestration (MAESTRO), supervision (KORTEX) and analytics (IMAGO) convert system complexity into coordinated control. Collectively, these elements ensure the resilience, compliance and sustainability of pharmaceutical manufacturing.
Why usability is a strategic requirement
Modern automation systems provide real-time optimisation, self-learning, and adaptive controls for flexible manufacturing. Automation, including AI, plays a vital role in the digital transformation of pharmaceutical production. Every day, the industry implements more advanced tools that improve machine performance, increase operational efficiency and help meet stricter regulations. At the same time, this rapid progress naturally introduces additional complexity. And here lies the challenge. As automation advances, it bears an important responsibility: innovation must not compromise usability. Ensuring that advanced automation remains accessible, intuitive and easy to understand is crucial for technicians, operators and engineers alike. Intelligent and adaptable systems will only truly support agile, reliable and sustainable pharmaceutical manufacturing as long as people are kept at the centre.
Within this context, DOMINA and SOLIDA, IMA Active’s latest generation tablet presses, represent this evolution. Their control architectures are intelligent and interconnected, providing consistent, real-time control while adding self-adapting and self-learning features. At the core of this challenge is a fundamental awareness: behind every system and machine are people. Their success depends directly on the design’s effectiveness. The goal is therefore to create simplicity within complexity, ensuring that advanced technologies remain usable and reliable while supporting daily operations.
This approach is demonstrated in the KORTEX interface, which offers tailored information based on each user’s skill level. It actively guides users through workflows and procedures that reduce errors, thus significantly reducing training time. At the same time, it provides deeper access to detailed information whenever a more thorough understanding of machine behaviour is needed, turning data into meaningful insights at any level.
By combining advanced architectures with clarity and continuous customer feedback, automation systems remain sophisticated yet usable, striking a balance between functionality and operational transparency. In this way, innovation does not add confusion but enhances usability and enables operators, technicians and engineers to work with confidence.
From Pharma 4.0 to a holistic, human-centric design (towards 5.0)
To achieve this balance between advanced automation and usability, we adopt a holistic design approach. But what does this mean in practice?
It is a design philosophy that recognises how pharmaceutical manufacturing operates within a broad, interconnected system. Mechanical design, software architecture, industrial engineering, process understanding, regulatory requirements and human interaction are not separate layers but parts of a single, unified whole.
These principles are embedded in well-known operating models such as Pharma 4.0 and Pharma 5.0, which encourage digital transformation, smart manufacturing and a strong focus on human factors. Specifically, Pharma 5.0 extends beyond digitalisation, emphasising the comprehensive integration of advanced technologies across all phases of pharmaceutical development and production.
The future of manufacturing is not a battle between humans and machines. It is a collaboration that promises to redefine productivity and innovation. Industry 5.0 is poised to revolutionise how companies operate over the next decade.1
Regulatory compliance has long been a critical pillar of automation design. Standards such as 21 CFR Part 11 and Annex 11 are non-negotiable. However, new and increasingly sensitive challenges are emerging. With the exponential growth of data, Data integrity must be addressed by design, not treated as an afterthought added late in the project lifecycle.
Embedding cybersecurity into automation is not merely a technical requirement — it is a commitment to patient safety, product quality and regulatory excellence”
In parallel, cybersecurity has become an area in which the full magnitude of the challenge is only now being understood. Embedding cybersecurity into automation is not merely a technical requirement — it is a commitment to patient safety, product quality and regulatory excellence.
A holistic automation strategy, therefore, aims to create a connected and interoperable ecosystem in which OT systems, such as PLCs and SCADA, seamlessly integrate with IT layers, including MES, ERP, and data analytics, as well as IoT devices, intelligent sensors and edge technologies. The objective is to develop a system that consistently supports documented quality, product reliability and long-term operational resilience.
The team and the methods behind the systems
Placing people at the centre begins with the team’s structure. System design is not just a task for software engineers or automation programmers. It requires incorporating diverse expertise — including UX designers, process specialists, compliance experts and ongoing customer engagement — supported by structured, continuous feedback loops.2
This approach reflects a growing consensus within Industry 5.0 and human-centred automation frameworks: effective systems emerge at the intersection of technology, organisation and people.3
Effective systems emerge at the intersection of technology, organisation and people”
In practice, this involves intentionally breaking down traditional silos between mechanical, automation and software design. A specific example is the use of digital twins during development. By modelling the physical system in real time, digital twins enable teams to identify usability issues, functional constraints and operational risks early in the design process, thereby informing decisions before implementation. When designed with the operator in mind, digital twins enhance human–machine collaboration rather than overwhelming users with complexity.4
Cybersecurity represents another critical dimension of this multidisciplinary effort. Current and forthcoming regulatory frameworks — including the NIS2 Directive and the EU Machinery Regulation 2023/1230, which take effect in 2027 — are reshaping the concept of safety by explicitly linking it to cybersecurity.5
Current and forthcoming regulatory frameworks… are reshaping the concept of safety by explicitly linking it to cybersecurity”
This shift reinforces the principle of ‘safety through security’, requiring cybersecurity considerations to be embedded throughout the machine lifecycle, from design to operation and maintenance.6
Addressing these requirements effectively calls for close cooperation among automation engineers, IT/OT cybersecurity specialists and compliance experts.
The team behind IMA Active reflects this strategic choice. It brings together software engineers, process specialists, UX designers and compliance experts, working in close coordination to ensure seamless interaction between machines and people. By integrating these perspectives into the design process, systems are developed that are not only technically advanced, but also understandable, trustworthy and effectively used in daily operations. This multidisciplinary model directly supports the human-centric principles described in Pharma 4.0 and Industry 5.0, translating them into concrete engineering practices.
Interfaces that guide, not hide
The SOLIDA family represents a benchmark as a reliable, efficient and cost‑effective tablet press, designed to meet the stringent requirements of pharmaceutical manufacturing. In this context, reliability, precision and consistency are not aspirational targets but operational necessities to ensure product quality, regulatory compliance and uninterrupted production.7
A robust machine architecture minimises downtime, reduces operational risk and delivers repeatable performance across batches, making SOLIDA a strategic investment rather than a simple equipment choice.
Although the automation platform is uniquely designed, it is intentionally balanced between informational detail and operational simplicity, tailored to the specific needs of daily manufacturing operations.8 The automation adapts to its environment, communicates clearly and supports confident decision-making, thereby enabling operators to act quickly and safely. Meanwhile, technical specialists have access to more detailed information when needed.
In pharmaceutical manufacturing, simplifying does not mean hiding information. On SOLIDA, the human–machine interface follows a top‑down design approach, providing immediate visibility of machine status and key performance indicators to maximise productivity, while allowing progressive drill‑down into detailed parameters and historical data. This approach is consistent with human‑centred HMI design principles, which aim to reduce cognitive load for operators without limiting the analytical depth available to expert users.9
A clear example of this philosophy is the sampling system. On the one hand, sampling operates fully automatically, ensuring consistent and reliable in‑process quality control without requiring manual operator intervention. The same system also provides immediate and direct feedback through a structured, top‑down approach. Technical specialists can examine every aspect of the sampling process in detail, including data acquisition, trends and outcomes, thereby gaining a comprehensive understanding of machine behaviour. This level of transparency enables informed decision‑making and continuous process optimisation, fully aligned with principles promoted by regulators for modern pharmaceutical manufacturing.10
PAT and QbD: from ‘tool’ to ‘sensor’
For continuous process optimisation, one of the most effective enablers is PAT. The pharmaceutical industry has historically been cautious about innovation, as validated processes tend to remain ‘frozen’ to minimise regulatory risk and avoid delays in product approval. However, innovation has been officially recognised by regulatory authorities as a key factor in process robustness, supply resilience and public health protection.11
For this reason, regulatory agencies have launched targeted initiatives to promote the voluntary adoption of innovative technologies, including PAT, by clarifying expectations and lowering perceived compliance barriers.11 In this context, QbD offers the strategic framework for process design. Simultaneously, PAT provides the analytical tools and real-time data needed to implement, sustain and enhance that strategy throughout the product lifecycle. This combination underpins continued process verification (CPV), real-time release testing (RTRT) and robust in-line quality control.
Without the ability to continuously monitor critical process parameters (CPPs) and critical quality attributes (CQAs) in real time, it would be impossible to keep a process dynamically within its design space in a proactive and controlled way. PAT, therefore, shifts quality assurance from retrospective verification to active, knowledge-based process control.
In this context, making PAT usable at the shop floor requires a conceptual shift —seeing it as an integrated ‘sensor’ instead of a specialised ‘tool’. This is not merely a semantic difference but a deliberate choice for improved usability. The aim is to present PAT-derived information with the same clarity and immediacy as traditional measurements such as temperature, force, or speed, even though the underlying analytical complexity is much greater.
Flexible manufacturing by design: DOMINA
The design of modern tablet press control systems must address the increasing need for flexibility and scalability across different production paradigms, including batch, semi-batch and continuous manufacturing.
A single dominant operating mode no longer defines pharmaceutical production; instead, manufacturers must be able to adapt rapidly to changing market demands, product lifecycles and regulatory expectations while maintaining consistent quality and compliance.12
Within this evolving landscape, the DOMINA family has been engineered with a specific forward-looking design focus, enabling seamless integration into continuous manufacturing (CM) environments. This approach reflects a deliberate strategic choice, recognising that flexibility across operating modes is essential to future-proof pharmaceutical manufacturing assets.
The continuous manufacturing paradigm offers well-documented advantages, including improved process efficiency, reduced production costs, enhanced product quality consistency, and faster time‑to‑market. However, these benefits come with inherent challenges. Continuous operations require robust process integration, real-time data availability and advanced control strategies.
A key element in addressing these challenges is the MAESTRO orchestration system, which integrates multiple machines, enabling the user to manage the entire manufacturing line as a single system, regardless of the machines and systems involved. Within this architecture, the DOMINA automation platform plays a pivotal role in supporting continuous manufacturing. It provides real‑time data exchange, adaptive control mechanisms and a scalable automation architecture that ensures flexibility and robustness across varying production demands.
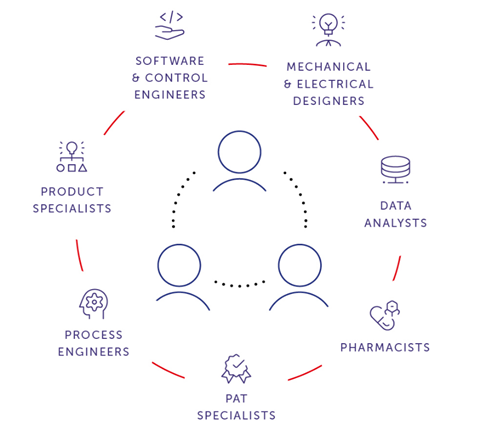
The automation ecosystem: KORTEX, MAESTRO, IMAGO
Referring to an automation ecosystem rather than to individual automation components underscores that system integration is a strategic design principle rather than a secondary implementation detail. In modern pharmaceutical manufacturing, value is generated not by isolated machines but by the coordinated interaction of supervision, orchestration and analytics layers that operate as a single, coherent system.8
In modern pharmaceutical manufacturing, value is generated not by isolated machines but by the coordinated interaction of supervision, orchestration and analytics layers that operate as a single, coherent system”
Within this ecosystem, each layer has a clearly defined role.
KORTEX provides machine supervision, with the interface enabling direct data interaction . Users can search and query data, and narrow in on trends to analyse details.
MAESTRO is an orchestration system that can also serve as a machine manager, centralising all supervisory activities — production scheduling, recipe editing, batch log management and data aggregation — to maximise machine availability.
IMAGO enables data analysis and interacts directly with machines to set specific test runs and extract the most valuable information.
This layered architecture reflects a fundamental shift in automation philosophy. As manufacturing systems become more connected and data-rich, the challenge is no longer data availability, but the ability to convert data into information, and information into action.8

The automation ecosystem: KORTEX, MAESTRO, IMAGO (Credit: IMA Active)
Sustainability: from efficiency to resilience
Sustainability in pharmaceutical manufacturing extends far beyond isolated efficiency measures or simple waste reduction initiatives. It requires a systemic approach that combines process optimisation, energy awareness and operational flexibility to build resilient manufacturing processes capable of ensuring a reliable supply of medicines over time. Sustainability is therefore inseparable from resilience, the ability of production systems to adapt, respond and continue operating effectively under changing market conditions and external constraints.
Sustainability in pharmaceutical manufacturing… requires a systemic approach that combines process optimisation, energy awareness and operational flexibility to build resilient manufacturing processes”
This framework means energy performance is a strategic lever rather than a secondary metric. Tools such as the Energy Diary enable real-time monitoring of energy consumption, providing clear, data-driven insights and a comprehensive view of energy usage across machines, lines and operations. By making energy data visible and actionable, manufacturers can better understand the full lifecycle impact of their processes, identify opportunities for optimisation, and support informed, long-term decision-making.
This approach allows efficiency to evolve from a purely economic objective into a core principle of responsible industrial practice. Efficient use of energy, materials and assets reduces environmental impact while simultaneously strengthening operational stability and long-term competitiveness. Such alignment among performance, resilience and sustainability is central to contemporary interpretations of Industry 5.0 and human-centric manufacturing.
The SOLIDA and DOMINA tablet press families illustrate this integrated approach. Through innovations in automation, control and system integration, they show how design choices can support flexible production, energy efficiency and resilient operations, helping shape a future-ready pharmaceutical manufacturing landscape where sustainability is built in from the start rather than added later.
Delivering a holistic future in manufacturing automation
Holistic automation integrates technology, people, processes and regulations into a single, coherent manufacturing strategy. As automation systems become more powerful and data-intensive, usability, data integrity and cybersecurity emerge as foundational design requirements rather than optional features. By embedding these principles from the earliest design stages, advanced control systems can remain transparent, trustworthy and effectively adopted on the shop floor.
IMA Active demonstrates how holistic automation turns complexity into control. By doing so, it strengthens operational resilience, supports sustainability objectives and allows manufacturing teams to focus on what matters most: reliable processes, consistent tablet quality and a robust supply of medicines over time.
References
1. Eschbach A. Beyond Automation: The Human Touch of Industry 5.0. [Internet] ISPE. 2024. Available from: https://ispe.org/pharmaceutical-engineering/ispeak/beyond-automation-human-touch-industry-50
2. Eriksson K, Olsson K, Danielsson F. Transdisciplinary Engineering and Co-Creation Towards Human‑Centric Smart Automation. [Internet] Advances in Transdisciplinary Engineering. 2024. Available from:
https://www.diva-portal.org/smash/get/diva2:1918835/FULLTEXT01.pdf
3. Pharma 4.0™: An Operating Model for Pharmaceutical Manufacturing. [Internet] ISPE. 2024. Available from: https://ispe.org/initiatives/pharma-4.0
4. Shahab, M. A, Destro F, Braatz D. Digital Twins in Biopharmaceutical Manufacturing: Review and Perspective on Human–Machine Collaborative Intelligence. [Internet] arXiv preprint. 2025. Available from: https://arxiv.org/abs/2504.00286
5. Regulation (EU) 2023/1230 on Machinery. [Internet] European Union. Official Journal of the European Union. 2023. Available from: https://eur-lex.europa.eu/eli/reg/2023/1230/oj
6. Directive (EU) 2022/2555 — Network and Information Security (NIS2). [Internet] European Union. 2022. Available from: https://eur-lex.europa.eu/eli/dir/2022/2555/oj
7. EudraLex — Volume 4: EU Guidelines for Good Manufacturing Practice. Annex 11 (Computerised Systems) & Annex 15 (Qualification and Validation). [Internet] European Commission. Available from: https://health.ec.europa.eu/medicinal-products/eudralex/eudralex-volume-4_en
8. ISPE Pharma 4.0™ Special Interest Group Digital Maturity, Operating Model and Organisational Enablers. [Internet] ISPE. 2024. Available from: https://ispe.org/initiatives/pharma-4.0
9. ISA‑101.01-2015— Human‑Machine Interfaces for Process Automation Systems. [Internet] ISA. Available from: https://www.isa.org/products/ansi-isa-101-01-2015-human-machine-interfaces-for?_gl=1*hwycs6*_up*MQ..*_ga*MTc3MzQwMjc4MC4xNzc0NjE3NTg1*_ga_8Z8VGE0R98*czE3NzQ2MTc1ODUkbzEkZzAkdDE3NzQ2MTc1ODUkajYwJGwwJGgxNTA3NDQwNTM2
10. ICH Q8(R2): Pharmaceutical Development. [Internet] ICH. 2009. Available from: https://database.ich.org/sites/default/files/Q8_R2_Guideline.pdf
11. PAT — A Framework for Innovative Pharmaceutical Development, Manufacturing, and Quality Assurance. FDA. 2004.
12. ISPE Pharma 4.0™ Initiative — Digital Transformation Framework.