Expanded EU and US approvals for Bristol Myers Squibb’s Opdivo (nivolumab) position combination therapy as frontline option for the common blood cancer.
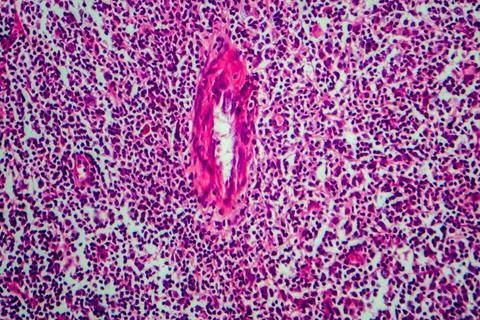
Biologic Opdivo (nivolumab) is now authorised as a combination therapy for two new classical Hodgkin Lymphoma indications.
Firstly, the FDA has authorised the monoclonal antibody alongside doxorubicin, vinblastine and dacarbazine (AVD) in individuals 12 years old and over with previously untreated, Stage III or IV cHL. It represents the first immunotherapy combination approved for patients with this indication.
The US authorisation is based on the phase III SWOG 1826 (CA2098UT) study. Data shows the combination treatment enabled a 58 percent reduction in the risk of disease progression or death.
Dr Alex Herrera, Chief of Division of Lymphoma, City of Hope National Medical Center, US, said: “In the US, the nivolumab-based combination for patients with previously untreated Stage III or IV cHL demonstrated improved progression-free survival compared with standard of care, BV-AVD. The SWOG 1826 study provides data for frontline use of this immunotherapy-based regimen.”
[Authorisation of Opdivo with brentuximab vedotin in the EU] represents a milestone as the first immunotherapy combination for certain [cHL] relapsed or refractory patients”
[Authorisation of Opdivo with brentuximab vedotin in the EU] represents a milestone as the first immunotherapy combination for certain [cHL] relapsed or refractory patients”
Alongside this, in the EU, Opdivo is now approved in combination with brentuximab vedotin for individuals between five and 30 years old with relapsed or refractory cHL following one prior line of therapy.
Dr Monica Shaw, Senior Vice President of Oncology Commercialisation, noted that this authorisation in the EU represents “a milestone as the first immunotherapy combination for certain relapsed or refractory patients”.
The European Commission’s decision is based on the phase II CheckMate -744 (CA209744) study. The combination treatment provided high complete metabolic response rates in children, adolescents, and young adults. Additionally, durable responses were observed at follow-up. These findings were presented at the 2023 American Society of Clinical Oncology (ASCO) Annual Meeting.
European Medicines Agency (EMA) is currently reviewing a regulatory submission based on SWOG 1826 trial.
Ono Pharmaceutical agreed that BMS could develop Opdivo globally following a collaboration agreement in 2011.
However, Bristol Myers Squibb is one of the companies expected to bear the brunt of the upcoming patent cliff as Opdivo, alongside its anticoagulant drug Eliquis (apixaban), are set to lose exclusivity in the US market over the next decade.
Sandoz recently agreed a new licensing deal with Samsung Bioepis to help it navigate this shift in the biosimilars market. It involves a candidate for the reference medicine Entyvio, used to treat Crohn’s disease.














No comments yet