All Drug Development articles – Page 16
-
 News
NewsNovel nanoemulsion-based delivery of antimycobacterial drug
The manufacturing process for the nanoemulsion could be adapted for industrial-scale production, the researchers suggested.
-
 News
NewsAbbVie agrees $1.4 billion Alzheimer's acquisition
The deal will help AbbVie to strengthen its neuroscience pipeline, including supporting development of next-generation therapies for delivery into the central nervous system.
-
 News
NewsACG 2024: J&J data reveals new Crohn's treatment option
More patients given subcutaneous induction and maintenance treatment with Johnson & Johnson’s biologic obtained clinical remission at 48 weeks, compared to placebo, Phase III data showed.
-
 News
NewsTrial highlights potential next-gen biologic for lung cancer
Preliminary results from an ongoing trial of a novel monoclonal antibody have confirmed favourable efficacy and safety in recurrent extensive-stage small cell lung cancer (SCLC).
-
 Article
ArticleDeveloping donanemab - balancing cost versus benefit
This article summarises recent regulatory developments surrounding the Alzheimer’s treatment donanemab and considers the future therapeutic market for this disease.
-
 News
NewsEli Lilly secures MHRA authorisation for Alzheimer's treatment
Authorisation in Great Britain means eligible Alzheimer's patients can access the treatment, which is part of a new class of amyloid targeting therapies.
-
 News
NewsWorld-first medicine manufacturing regulatory framework to be implemented
The new regulation could become lawful in the UK next year, according to the Medicines and Healthcare products Regulatory Agency (MHRA).
-
 News
NewsDrug combination shows promise for mitigating microbial biofilms
Combining allicin with domiphen could reduce biofilm formation, providing a potential avenue for overcoming antimicrobial resistance, research suggests.
-
 News
NewsImmunotherapy treatment enhances lymphoma survival, landmark study shows
The Phase III findings give evidence for the immunotherapy treatment regimen to become standard of care for advanced Hodgkin lymphoma.
-
 News
NewsSanofi collaboration to advance innovative radioligand therapies
The new partnership between Sanofi and Orano Med, delivered via a new entity valued at nearly $2 billion, will focus on progressing lead-212 based targeted alpha therapies for oncology indications.
-
 Article
ArticleInnovating allergy drug delivery with a needle-free alternative
Dr Sarina Tanimoto, MBA, Co-Founder and Chief Medical Officer of ARS Pharmaceuticals, discusses the company’s EURneffy adrenaline nasal spray as a novel, needle-free option to older approved injectables such as intramuscular injection, following its authorisation in the EU and US.
-
 News
NewsHaemophilia treatment with new delivery method approved
The US FDA has authorised the first treatment for haemophilia A or B that is administered via a pre-filled pen or syringe.
-
 News
NewsEMA publishes EU strategy to 2028
The draft strategy, open for an eight-week public consultation, will help guide the direction of the network over the next few years.
-
 News
NewsEli Lilly appoints Chief AI Officer
The new appointee will create AI and machine learning solutions to support Eli Lilly’s medicines for patients globally.
-
 Whitepaper
WhitepaperA data-driven approach to Container Closure Integrity
Selecting container closure components with confidence:A data-driven approach to Container Closure Integrity.
-
 Article
ArticleCPHI Milan 2024: everything you need to know
CPHI Milan takes place in October, covering a wide range of topics from packaging and drug delivery to integrated pharma.
-
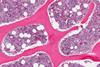 News
NewsFirst real-world study reveals encouraging efficacy for cell therapy
More than half of the multiple myeloma patients included in this study would have been ineligible for the Phase II CARTITUDE-1 clinical trial, research reports.
-
 News
NewsEnhancing intestinal drug delivery with Quality by Design
Research has highlighted a potential oral drug delivery approach using Quality by Design (QbD) principles to improve treatment of the colonic disease diverticulitis.
-
 Article
ArticleFuture of oral small molecule drugs - addressing potential in multiple sclerosis
Advances in selectivity, safety, and patient convenience, oral small molecule drugs continue to be a key focus in drug development. Here, Dr Andreas Muehler and Daniel Vitt, PhD, the co-founders of Immunic Therapeutics, discuss the potential of Vidofludimus calcium as a breakthrough treatment for autoimmune diseases.
-
 Article
ArticleQuality from the start: designing a scalable AAV production platform
This Feature Partnership discusses AAVs and how manufacturers can overcome the key development challenges and create a scalable AAV production platform.



