FDA approves Fasenra for severe eosinophilic asthma
Posted: 16 November 2017 | Dr Zara Kassam (European Pharmaceutical Review) | No comments yet
The FDA has approved Fasenra for the add-on maintenance treatment of patients with severe asthma aged 12 years and older…

AstraZeneca and its global biologics research and development arm, MedImmune, has announced that the US Food and Drug Administration (FDA) has approved Fasenra (benralizumab) for the add-on maintenance treatment of patients with severe asthma aged 12 years and older, and with an eosinophilic phenotype.
Fasenra has a strong clinical profile which includes the ability to show lung function improvement after the first dose
Pascal Soriot, Chief Executive Officer of AstraZeneca, said: “We’re excited to offer Fasenra as a new precision biologic to help improve the lives of severe asthma patients whose disease is driven by eosinophilic inflammation. This is the first approval from our respiratory biologics portfolio and the latest in a series of significant milestones for our company as we deliver on our pipeline-driven transformation.”
The FDA approval is based on results from the WINDWARD programme, including the pivotal Phase III exacerbation trials, SIROCCO and CALIMA, and the Phase III oral corticosteroid (OCS)-sparing trial, ZONDA. Results for the 8-week benralizumab dosing regimen from these trials showed:
- Up to 51% reduction in the annual asthma exacerbation rate (AAER) versus placebo
- Significant improvement in lung function as measured by forced expiratory volume in one second (FEV1) of up to 159mL versus placebo. Differences were seen as early as 4 weeks after the first dose, providing an early indication of effectiveness
- 75% median reduction in daily OCS use and discontinuation of OCS use in 52% of eligible patients
- An overall adverse event profile similar to that of placebo.
Dr Eugene Bleecker, Professor and Co-Director, Genetics, Genomics and Precision Medicine, University of Arizona Health Sciences, and lead investigator of the pivotal Phase III SIROCCO study, said: “This is an important day for severe, eosinophilic asthma patients who have had limited treatment options for far too long, with many relying on oral steroids to manage their symptoms. Fasenra has a strong clinical profile which includes the ability to show lung function improvement after the first dose, the potential to reduce – or even stop – oral steroid use, and the convenience of 8-week dosing. Fasenraalso treats a distinct patient phenotype, helping physicians select the right patient in clinical practice with more confidence.”
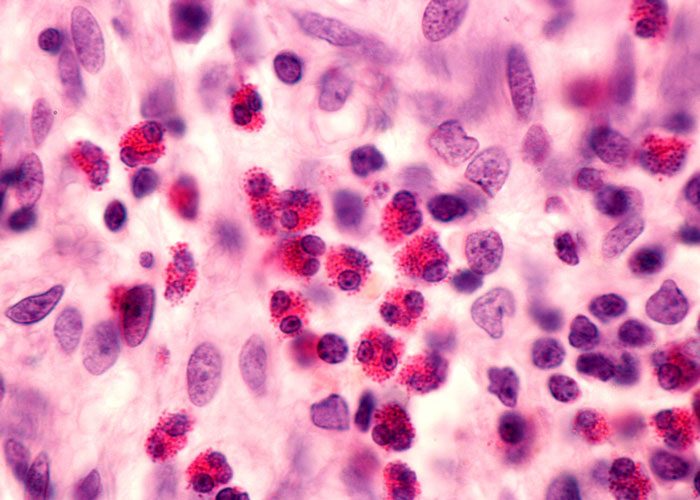
Fasenra is the only respiratory biologic that provides direct, rapid and near-complete depletion of eosinophils within 24 hours. Eosinophils are a type of white blood cell that are a normal part of the body’s immune system. Elevated levels of eosinophils, seen in about half of severe asthma patients, impact airway inflammation and airway hyper-responsiveness, resulting in increased asthma severity and symptoms, decreased lung function and increased risk of exacerbations.
Fasenra binds directly to the IL-5α receptor on an eosinophil and uniquely attracts natural killer cells to induce apoptosis. Fasenra will be available as a once every 8-week fixed-dose subcutaneous injection via a prefilled syringe.
On 10 November 2017, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) adopted a positive opinion recommending the marketing authorisation of benralizumab. Benralizumab is also under regulatory review in Japan and several other countries.
About Fasenra
Fasenra is a monoclonal antibody that recruits natural killer cells to induce direct, rapid and near-complete depletion of eosinophils. Depletion of circulating eosinophils is rapid, with an onset of action within 24 hours as confirmed in an early Phase II trial. In the pivotal Phase III trials, SIROCCO and CALIMA, Fasenra demonstrated a significant reduction in exacerbations and improved lung function and asthma symptoms in severe, uncontrolled eosinophilic asthma patients.
Eosinophils are the biological effector cells in approximately 50% of asthma patients, leading to frequent exacerbations, impaired lung function and asthma symptoms. Fasenra will be available as a subcutaneous injection via a prefilled syringe administered once every 4 weeks for the first 3 doses, and then once every 8-weeks thereafter.
Fasenra is now approved in the US, and under regulatory review in the EU, Japan and several other countries.
Fasenra is the foundation of AstraZeneca’s respiratory biologics portfolio of potential new medicines targeting underlying causes of respiratory disease. Fasenra is also being evaluated in chronic obstructive pulmonary disease (COPD).
Fasenra was developed by AstraZeneca with MedImmune, AstraZeneca’s global biologics research and development arm, and is in-licensed from BioWa, Inc., a wholly-owned subsidiary of Kyowa Hakko Kirin Co., Ltd., Japan.
Related topics
Related organisations
AstraZeneca, MedImmune, U.S. Food and Drug Administration (FDA)