Small molecule mediators of cell differentiation: Tools to generate tissue from stem cells for screening applications
Posted: 3 December 2008 | Dr Stefan Przyborski, Chief Scientific Officer, Reinnervate Limited, School of Biological and Biomedical Science, Durham University | No comments yet
The development of pharmaceuticals and screening the biological activity of test compounds is a multi-staged process spanning from small molecule design and synthesis, in vitro testing, and compound evaluation in vivo using animal and human trials. The expense of this process escalates as a compound advances further into the development programme. Within each stage there are stringent criteria that must be satisfied before the decision to invest further in the compound and move its development forward is made.
The development of pharmaceuticals and screening the biological activity of test compounds is a multi-staged process spanning from small molecule design and synthesis, in vitro testing, and compound evaluation in vivo using animal and human trials. The expense of this process escalates as a compound advances further into the development programme. Within each stage there are stringent criteria that must be satisfied before the decision to invest further in the compound and move its development forward is made.
The development of pharmaceuticals and screening the biological activity of test compounds is a multi-staged process spanning from small molecule design and synthesis, in vitro testing, and compound evaluation in vivo using animal and human trials. The expense of this process escalates as a compound advances further into the development programme. Within each stage there are stringent criteria that must be satisfied before the decision to invest further in the compound and move its development forward is made.
The pharmaceutical industry is continually reviewing its practices and seeking novel ways in which to bring new drugs to market in a more efficient and cost effective manner. Part of this process will be to avoid investment in a potential drug candidate that fails at a later stage in the development process. The trick of course is spotting when to stop research and development of a particular candidate but at the same time to learn from the experience.
Investment in research tools and procedures at the early stages of product development could yield significant cost savings. One area that holds much promise is the use of stem cells as a consistent supply of tissues for in vitro testing of drug candidates. Stem cells provide a renewable source of tissue. They can self-renew and differentiate into cells of alternative phenotypes. There are various types of stem cell, derived from different sources, including embryonic, foetal and adult tissues. Depending on the type of stem cell will determine its development potential or potency to differentiate into a range of alternative cell types. Embryonic stem cells are referred to as being totipotent since they retain the capacity to differentiate into all tissues of the body, whereas adult stem cells are understood to be more restricted in their developmental potential. One of the classical ways to demonstrate the developmental potential of embryonic stem cells is to transplant them into an immune deficient host and grow a teratoma1. A teratoma is a form of tumour consisting of a haphazard array of differentiated tissue types arising from the grafted stem cells.
Histological examination of the structure of a teratoma reveals the enormous potential of stem cells to produce tissues for use in research and development. In addition, there is the ability to work with stem cells derived from human material, thus enabling researchers to study the biological activity of test molecules in a system directly relevant to man. Indeed, the use of human stem cells in research and regenerative medicine has become especially topical since the isolation of human embryonic stem cells by Thomson and colleagues in 19982. In terms of drug development, the use of human cells has the added advantage of identifying any inter-species differences and toxicity issues that could be missed working in animal derived systems.
A major challenge in stem cell biology is the ability to control the development of cells in a predicable way and produce tissues of a particular type in a robust and reproducible manner. Cultured embryonic stem cells and progenitor cells from the early embryo have the potential to recapitulate the early stages of embryogenesis, making them a valuable tool to research the molecular mechanisms that regulate development. Decades of research by developmental biologists have focused on studying such pathways in various model systems, ranging from worms, flies, fish and mice. Hence a significant body of knowledge has accumulated on the signalling pathways involved in the control of cell differentiation. More recently, scientists are applying such knowhow to cultured stem cells in an attempt to recapitulate the early stages of embryogenesis and coax them toward differentiating into specific cell types. In turn such cells can be used as a consistent source of tissue for testing drugs and evaluating compound toxicity.
To aid this process, cell culture procedures that induce reliable and reproducible cell differentiation resulting in consistent populations of differentiated cells are needed. This is especially important where reproduction of cell behaviour is required, for example, the consistent differentiation of neurons from stem cells for use in culture tests and drug development. Currently, this is not always easy to achieve since there are discrepancies between in vitro protocols and inconsistency in reagents used to modulate cell differentiation. Researchers require a broad portfolio of reliable ‘off-the-shelf’ reagents and protocols that interact with specific molecular pathways in order to control the process of cell differentiation in a predictable manner. These reagents should be designed to enable a specific process and overcome a source of variability resulting in enhanced cell behaviour and consistency.
The following example is used to highlight this issue. Retinoids are naturally occurring derivatives of vitamin A (retinol). All-trans-retinoic acid (ATRA) is involved in the control of many biological processes, both during embryonic development and in adult life, particularly in nervous tissues3. Retinoids are excellent chromophores which efficiently absorb light in the region of 300-400 nm (depending on the solvent). Exposure to light, and other factors such as temperature and oxidation, makes these molecules particularly susceptible to isomerisation, leading to degradation into a mixture of retinoic acid isomers. Compound stability is an important feature when reagents are routinely used for cell culture applications. During the preparation and maintenance of cell cultures, the concentration levels of retinoids have been shown to decrease markedly over time, and this could be a direct consequence of both their degradation and metabolism. Isomerisation of ATRA is understood to be an important part of its metabolic pathways within cells, since the resulting isomers have different mechanisms of action. Indeed, this is an important point that is all too often over-looked by users of natural retinoids in the cell culture laboratory. A warning to this effect has been issued to cell biologists using retinoic acids as inducers of cell differentiation by Murayama and co-workers (1997)4 who reported that the isomers of ATRA differentially affect the ability of mammalian stem cells to differentiate along alternative lineages, and they stated that care should be taken to protect retinoic acids from isomerisation. This is particularly relevant when the cellular response is determined by the concentration(s) of the isomer(s) present in solution. For example, the induction of the differentiation of pluripotent stem cells using retinoids is variable, resulting in the differential activation of key molecular pathways involved in tissue development in a concentration dependent manner5. In turn, this variation has the potential to result in mixed proportions of alternative differentiating cell types which leads to increased culture heterogeneity. To reduce such variability in differentiation response and improve reproducibility, it is essential that whatever is used to induce cell differentiation is in the same form and concentration every time it is used. Currently, this cannot be guaranteed when using reagents such as ATRA and its stereoisomers because they are highly susceptible to isomerisation under sample preparation conditions, storage of stock solution conditions and in use during culture.
To address this problem, we formed a collaborative alliance between biologists (Drs Przyborski and Hunt) and chemists (Dr Whiting and Professor Marder) at Durham University, UK. We have designed and synthesised a small collection of well-defined synthetic analogues of retinoic acid that possess biological activity similar to their native counter-parts with the added benefit of compound stability. An overview of this compound development process is summarised in Figure 1.
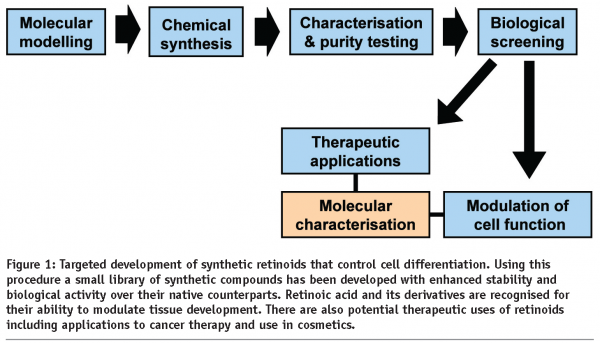

Retinoids are polyene-containing natural products and it is the combination of olefin geometries for these types of compound that has a profound effect on biological processes. We are developing methods for the stereo-controlled synthesis of polyene building blocks from which it will assemble retinoid-related polyenes for biological testing (see Figure 2).
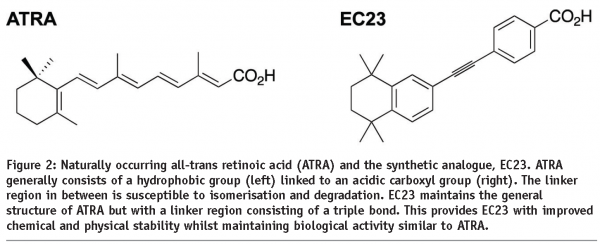

Rather than use a compound library screening approach, we studied the structure of the retinoid receptors and molecules known to interact with these receptors. Compound structures were modelled in silico prior to developing methods for their chemical synthesis. This targeted approach was successful and low numbers of molecules were produced which was amenable for screening the response to differentiating stem cells. Subsequent to their synthesis and purification, the chemical and physical stability of molecules was characterised by nuclear magnetic resonance. ATRA is often included in cell culture media formulations as a potent inducer of cell differentiation, particularly during the formation of neural derivatives. Samples of novel synthetic retinoids were therefore compared against ATRA for their ability to induce neurogenesis from stem cells.
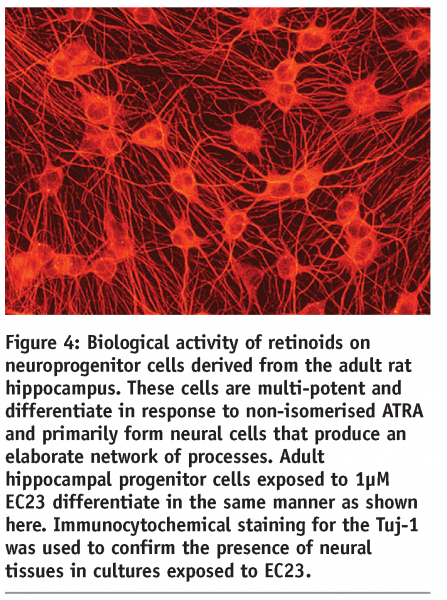
The outcome of this programme has been the development of a series of synthetic retinoid analogues that display improved chemical and physical stability and mimic the biological activity of naturally occurring ATRA. Two compounds in particular, EC23 and EC19, are the subject of a recently published research article5. These molecules are closely related in their structure: in EC23 the carboxyl group is located in the para position, whereas in EC19 the carboxyl group is located in the meta position. This is the only structural difference between the two molecules. Both EC23 and EC19 maintain structural integrity when exposed to intense light for 72 hours, whereas, ATRA photo-isomerises within minutes. We have demonstrated the application of this technology by testing the ability of these retinoids compounds to induce the differentiation of mammalian stem cells. In each case the biological activity of the synthetic retinoid was compared to that of naturally occurring ATRA. We have generated extensive data showing that EC23 is a potent inducer of neural differentiation (see Figures 3 and 4).
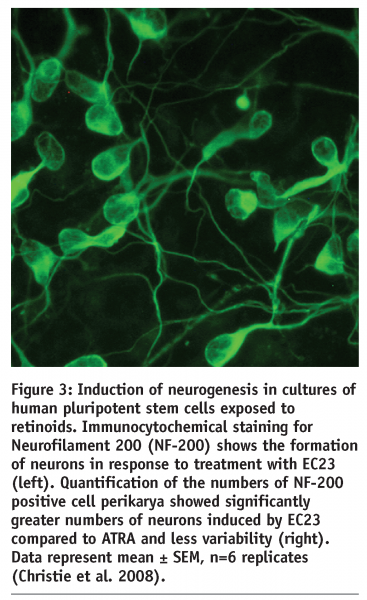

Human pluripotent embryonic stem cells and embryonal carcinoma stem cells exposed to EC23 readily differentiate and form neural derivatives. Similarly, neuroprogenitor cells derived from adult rat hippocampus form neural derivatives. Experimental data indicate that EC23 acts similarly to ATRA, however, EC23 does appear to be more potent than ATRA and can induce greater numbers of neurons in a more consistent manner (see Figure 3)5. In comparison, EC19 has little effect on the induction of neurogenesis by pluripotent stem cells and induces primarily non-neural epithelial cell types. Similarly, EC23 and EC19 possess differential effects on tissue development in vivo. Resin beads soaked in ATRA and implanted into the limb bud of the chick embryo can induce duplication of the limb. EC23 mimics the action of ATRA in this chick model but EC19 does not and induces a specific reduction in the frontonasal mass. In summary, these molecules are powerful tools for the modulation of cell differentiation and tissue development.

Subtle differences in the molecular structure can have dramatic effects on compound stability and on biological function, as exemplified above by the isomeric synthetic retinoids EC23 and EC19. Such information regarding structure-activity-relationships will be important for the future design and application of new synthetic compounds aimed at controlling cellular developmental processes. The observed similarity in the function of EC23 to ATRA highlights that the para-isomer synthetic retinoid is an exciting new compound for use in stem cell science since it maintains activity as a potent inducer of neurogenesis and has the added benefit of compound stability.
Finally, it is critical that the synthesis of such molecules can be readily adapted for manufacture on a sufficient scale to acceptable quality standards to deliver adequate supply to the market. Often new compounds are developed and reported but they are not readily accessible for use by the scientific community. We have demonstrated scaled manufacture of EC23 which is now available as a commercial product.
With careful design and thorough exemplification, new small molecules developed for specific applications in mediating the control of stem cell differentiation, will prove to be useful tools contributing to the provision of stem cell-derived tissues for use in screening and drug development. These technologies are necessary, together with the development of standard operating procedures to enable robust and consistent differentiation in a controlled manner. Using such tools, researchers will be able to reproducibly generate human cells and tissues from stem cells and progenitor cells as a reliable and renewable resource. Investment in such technology will ultimately lead to less variability, it will radically improve the effectiveness of in vitro assays, and the production of more meaningful data especially in human systems. Moreover, it is likely to have a significant impact on the success and overall efficiency of pharmaceutical development including potential cost savings, a reduction in the use of animals and the speed at which drug development can advance.
References
- Przyborski SA. (2005). Differentiation of human embryonic stem cells following transplantation into immune deficient mice. Stem Cells, 23, 1242-1250.
- Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM. (1998). Embryonic stem cell lines derived from human blastocysts. Science. 282, 1014-1015.
- Christie VB, Marder TB, Whiting A, Przyborski SA. (2008). Role of retinoids in the adult nervous system and their therapeutic potential. Mini Reviews in Medicinal Chemistry, 8, 601-608.
- Murayama A, Suzuki T, Matsui M. (1997). Photoisomerization of retinoic acids in ethanol under room light: a warning for cell biological study of geometrical isomers of retinoids. J Nutr Sci Vitaminol, 43, 167-176.
- Mavilio F, Simeone A, Boncinelli E, Andrews PW. (1988). Activation of four homeobox gene clusters in human embryonal carcinoma cells induced to differentiate by retinoic acid. Differentiation, 37, 73-79.
- Christie VB, Barnard JH,] Bridgens CE, Batsanov AS, Cartmell EB, Collings JC, Maltman DJ, Marder TB, Redfern CPF, Przyborski SA, Whiting AP. (2008). Synthesis and evaluation of synthetic retinoid derivatives as inducers of stem cell differentiation. Organic and Biomolecular Chemistry, 6, 3497-3507.





