Novel antisense drugs for micro-RNA therapeutics
Posted: 20 July 2006 | | No comments yet
MicroRNAs (miRNAs) are an abundant class of short endogenous RNAs that act as important post-transcriptional regulators of gene expression.
MicroRNAs (miRNAs) are an abundant class of short endogenous RNAs that act as important post-transcriptional regulators of gene expression.
MicroRNAs (miRNAs) are an abundant class of short endogenous RNAs that act as important post-transcriptional regulators of gene expression.
By base-pairing to their target mRNAs, they mediate cleavage of the mRNA or translational repression (Bartel 2004). To date nearly 4000 microRNAs have been annotated in vertebrates, invertebrates and plants according to the miRBase microRNA database release 8.1 in May 2006 (Griffith-Jones et al. 2006). Growing evidence suggests that miRNAs exhibit a variety of crucial regulatory functions related to cell growth, development and differentiation. Computational data combined with experimental analyses indicate that the total number of miRNAs in vertebrate genomes may be as many as 1000 (Bentwich et al. 2005, Berezikov et al. 2005). Recent bioinformatic predictions of miRNA targets in vertebrates indicate that microRNAs could be responsible for regulating up to 30% of the human protein-coding genes with an average of 200 target sites per miRNA (Krek et al. 2005, Lewis et al. 2005, Xie et al. 2005).
MicroRNAs in human disease
The expanding inventory of human microRNAs in addition to their highly diverse expression patterns and their role in many different cellular processes suggest that miRNAs are involved in a variety of human diseases. A mutation in the target site of miR-189 in the human SLITRK1 gene was recently shown to be associated with Tourette’s syndrome (Abelson et al. 2005), while another recent study reported that the hepatitis C virus (HCV) RNA genome interacts with a host-cell microRNA, the liver-specific miR-122a, to facilitate its replication in the host (Jopling et al. 2005). Perturbed miRNA ex-pression patterns have also been reported in many human cancers. For example, the human miRNA genes miR15a and miR16-1 are deleted or down-regulated in the majority of B-cell chronic lymphocytic leukemia (CLL) cases (Calin et al. 2002). The role of miRNAs in cancer is further supported by the fact that more than 50 per cent of the human miRNA genes are located in cancer-associated genomic regions or at fragile sites (Calin et al. 2004). Recently, systematic expression analysis of a diversity of human cancers revealed a general down-regulation of miRNAs in tumours, compared to normal tissues (Lu et al. 2005). Interestingly, miRNA-based classification of poorly differentiated tu-mours was successful, whereas mRNA profiles were highly inaccurate when applied to the same samples. miRNAs have also been shown to be dysregulated in breast cancer (Iorio et al. 2005), lung cancer (Johnson et al. 2005) and colon cancer (Michael et al. 2004), while the miR-17-92 cluster, which is amplified in human B-cell lymphomas and miR-155 which is upregulated in Burkitt’s lymphoma have been reported as the first human miRNA oncogenes (Eis et al. 2005, He et al. 2005).
LNA (locked nucleic acid) – a synthetic RNA mimick for high-affinity miRNA targeting
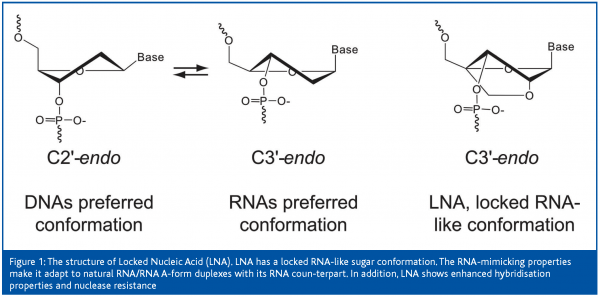
LNAs comprise a new class of nuclease-resistant bicyclic high-affinity RNA analogues in which the furanose ring in the sugar–phosphate backbone is chemically locked in an RNA mimicking N-type (C3’-endo) conformation by the introduction of a 2’-O,4’-C methylene bridge (Fig. 1; Koshkin et al., 1998). Several studies have demonstrated that LNA-modified oligonucleotides exhibit unprecedented thermal stability when hybridised with their RNA target molecules (Koshkin et al., 1998, Braasch and Corey, 2001; Kurreck, 2002). Consequently, an increase in melting temperature (Tm) of + 2-10°C per monomer against complementary RNA compared to unmodified duplexes have been reported. LNA incorporation generally improves mismatch discrimination compared to unmodified reference oligonucleotides. It should be underlined that LNA mediates high-affinity hybridisation without compromising base pairing selectivity, and that the standard Watson-Crick base pairing rules are obeyed.
The high thermal stability of short LNA oligonucleotides, together with their improved mismatch discrimination has facilitated the design and utility of LNA-modified DNA oligonucleotides in microRNA detection and analysis. In a recent paper, Valoczi et al., (2004) describe highly efficient detection of miRNAs by northern blot analysis using LNA-modified oligonucleotide probes and demonstrate their significantly improved sensitivity in detection of different miRNAs in mouse, Arabidopsis thaliana and Nicotiana benthamiana. The sensitivity in detecting mature microRNAs by northern blots was in-creased by at least ten-fold compared to end-labelled DNA probes of the same sequence, while simul-taneously retaining or even improving specificity (Valoczi et al. 2004).
The detection of mature miRNAs by in situ hybridisation has been technically challenging due to their small size. Recently, the spatial and temporal expression patterns of 115 conserved vertebrate miR-NAs were determined in zebrafish embryos and larvae directly by whole-mount in situ hybridisations using LNA-modified DNA probes similar to those used for northern blots (Wienholds et al. 2005). While most miRNAs were not detected in the early stages of development, at later stages they showed remarkably tissue-specific expression patterns, often limited to single organs or even single cell layers within an organ. The in situ patterns correlated well with zebrafish miRNA array data and with available miRNA expression data for mammals. The temporal expression patterns suggested that miRNAs may not directly act as developmental switches, but are involved in the maintenance of tissue identity (Wienholds et al. 2005).
In a more recent study, Kloosterman et al. (2006) describe a detailed analysis on the conditions for LNA probe-based in situ detection of miRNAs in zebrafish embryos, and demonstrate the utility of the method in Xenopus and mouse embryos. Using LNA-modified probes for miR-206, miR-124a and miR-122a in 72h-old zebrafish embryos revealed expected patterns for all three miRNAs, whereas no in situ signals could be deteted with cor-responding DNA probes, in accordance with the significantly increased affinity of LNA oligonucleotides toward complementary RNA molecules. Interestingly, specific detection of miR-206 and miR-124a could be achieved using shortened LNA versions comple-mentary to a 14-nt region at the 5′-end of the miRNA, thereby improving their specificity (Kloosterman et al. 2006). The successful use of very short probes with Tm values in the range of body tem-peratures has significant implications for therapeutic applications, such as inhibition of disease-related miRNAs.
Inhibition of microRNA function using LNA-antimiRs
Given their highly diverse biological functions and suggested roles in disease, human microRNAs have rapidly emerged as potential targets for therapeutic intervention. An effective strategy to silence miRNAs has been reported (Hutvagner et al. 2004; Leaman et al. 2005), in which 2’-O-methyl antisense oligonucleotides were used as potent and irreversible inhibitors of siRNA and miRNA function in vitro and in vivo in Drosophila and C. elegans, thereby inducing a loss-of-function phenotype. This method was recently applied to mouse studies, by conjugating 2’-O-methyl antisense oligonucleotides complementary to four different miRNAs with cholesterol (so called antagomirs) for silencing miRNAs in vivo (Krützfedt et al. 2005).
Recent studies have reported that LNA-modified oligonucleotides can also mediate specific inhibition of miRNA function (Chan et al. 2005; Lecellier et al. 2005; Ørom et al. 2006). Knockdown of miR-21 using LNA/DNA-mixed antisense oligonucleotides in cultured glioblastoma cells triggered activation of caspases and lead to increased apoptotic cell death (Chan et al. 2005). In this study, LNA antisense inhibitors were used in parallel with the 2’-O-methyl-oligonucleotides to inhibit miR-21 showing similar efficacy and specificity in the same range of concentrations when transfected with the same type of cationic liposomes (Chan et al. 2005).
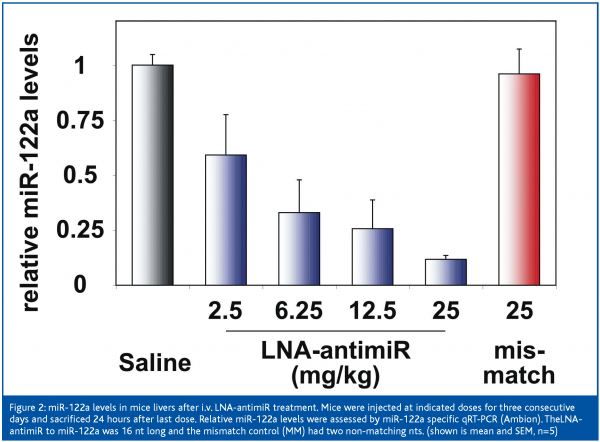
LNA oligonucleotides are readily transfected into cells using standard techniques, they are sequence-specific and non-toxic, and show good biodistribution and high serum stability in vivo, which make them highly useful for potent and selective antisense-based gene silencing (Kurreck et al 2002; Frieden et al. 2003, Wahlestedt et al. 2000; Braasch et al. 2002; Fluiter et al. 2003). Interestingly, LNA-modified DNA oligonucleotides show improved antisense efficacy and higher Tm toward comple-mentary RNA, compared to isosequential 2’-OMe-modified oligonucleotides (Kurreck et al. 2002, Grünweller et al. 2003), which have found wide use as miRNA inhibitors. These findings, along with the improved miRNA recognition properties of LNA in vitro, prompted us to assess the efficacy of LNA-antimiRs in miRNA inhibition in vivo. For this purpose we designed a 16-mer LNA-antimiR comple-mentary to the liver-specific miR-122a in mouse. NMRI mice were administered intravenously with LNA antimiR-122a using a dosing regimen ranging from 2.5 to 25 mg/kg for three consecutive days. Subsequent analysis of miR-122a levels in the harvested livers from LNA antimiR-122a treated mice by qRT-PCR revealed a dose-dependent reduction of the target miRNA with an IC50 of ca. 5-6 mg/kg compared to the saline-treated control mice (Figure 2). Taken together, our results suggest that LNA-substituted oligonucleotides are highly effective as miRNA inhibitors in vivo and, thus, represent an interesting, new class of microRNA antagonists with future potential in therapeutic intervention of dis-ease-associated microRNAs.


References
Abelson, J.F., Kwan, K.Y., O’Roak, B.J., Baek, D.Y., Stillman, A.A., Morgan, T.M., Mathews, C.A., Pauls, D.L., Rasin, M.R., Gunel, M., Davis, N.R., Ercan-Sencicek, A.G., Guez, D.H., Spertus, J.A., Leckman, J.F., Dure, L.S. 4th, Kurlan, R, Singer, H.S., Gilbert, D.L., Farhi, A., Louvi, A., Lifton, R.P., Sestan, N., State, M.W. 2005. Sequence variants in SLITRK1 are associated with Tourette’s syn-drome. Science 310: 317-20.
Bartel, D.P. 2004. MicroRNAs: Genomics, biogenesis, mechanism and function. Cell 116: 281-297.
Bentwich, I., Avniel, A., Karov, Y., Aharonov, R., Gilad, S., Barad, O., Barzilai, A., Einat, P., Einav, U., Meiri, E., Sharon, E., Spector, Y. and Bentwich, Z. 2005. Identification of hundreds of conserved and nonconserved human microRNAs. Nat. Genet. 37: 766–770.
Berezikov, E., Guryev, V., van de Belt, J., Wienholds, E., Plasterk, R.H.A. and Cuppen, E. 2005. Phylogenetic shadowing and computational identification of human microRNA genes. Cell 120: 21-24
Braasch, D.A. and Corey, D.R. 2001. Locked nucleic acid (LNA): fine-tuning the recognition of DNA and RNA. Chem Biol. 8: 1-7.
Braasch, D.A., Liu,Y. and Corey, D.R. 2002. Antisense inhibition of gene expression in cells by oli-gonucleotides incorporating locked nucleic acids: effect of mRNA target se-quence and chimera de-sign. Nucleic Acids Res. 30(23): e 5160-5167.
Calin, G. A., Sevignani, C., Dumitru, C.D., Hyslop, T., Noch, E., Yendamuri, S., Shimizu, M., Rattan, S., Bullrich, F., Negrini, M. and Croce, C.M. 2004. Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc. Natl. Acad. Sci.U.S.A. 101: 2999-3004.
Calin, G.A., Dumitru, C.D., Shimizu, M., Bichi, R., Zupo, S., Noch, E., Aldler, H., Rattan, S., Keating, M., Rai, K., Rassenti, L., Kipps, T., Negrini, M., Bullrich, F. and Croce, C.M. 2002. Frequent deletions and down-regulation of microRNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc. Natl. Acad. Sci. USA 99: 15524– 15529.
Chan, J.A., Krichevsky, A.M., Kosik, K.S. 2005. MicroRNA-21 is an antiapoptotic factor in human glioblastoma cells. Cancer Res. 65:6029-33.
Eis, P.S., Tam, W., Sun, L., Chadburn, A., Li, Z., Gomez, M.F., Lund, E., Dahlberg, J.E. 2005. Accu-mulation of miR-155 and BIC RNA in human B cell lymphomas. Proc Natl Acad Sci U S A. 102: 3627-32.
Fluiter, K., ten Asbroek, A.L.M., de Wissel, M.B., Jakobs, M.E., Wissenbach, M., Olsson, H., Olsen, O., Oerum, H. and Baas, F. 2003. In vivo tumor growth inhibition and biodistribution studies of locked nucleic acid (LNA) antisense oligonucleotides. Nucleic Acids Res. 31: 953-962.
Griffiths-Jones, S., Grocock, R.J., van Dongen, S., Bateman, A., Enright, A.J. 2006. miR-Base: mi-croRNA sequences, targets and gene nomenclature. Nucleic Acids Res. 34: D140-4
He, L., Thomson, J.M., Hemann, M.T., Hernando-Monge, E., Mu, D., Goodson, S., Pow-ers, S., Cor-don-Cardo, C., Lowe, S.W., Hannon, G.J. and Hammond, S.M. 2005. A microRNA polycistron as a potential human oncogene. Nature 435: 828– 833.
Hutvágner, G., Simard, M.J., Mello, C.C. and Zamore, P.D. 2004. Sequence-specific inhibition of small RNA function. PLoS Biology 2: 1-11.
Iorio, M.V., Ferracin, M., Liu, C.G., Veronese, A., Spizzo, R., Sabbioni, S., Magri, E., Pedriali, M., Fab-bri, M., Campiglio, M., Menard, S., Palazzo, J.P., Rosenberg, A., Musiani, P., Volinia, S., Nenci, I., Calin, G.A., Querzoli, P., Negrini, M., Croce, C.M. 2005. MicroRNA gene expression deregulation in human breast cancer. Cancer Res. 65: 7065-70.
Johnson, S.M., Grosshans, H., Shingara, J., Byrom, M., Jarvis, R., Cheng, A., Labourier, E., Reinert, K.L., Brown, D. and Slack, F.J. 2005. RAS is regulated by the let-7 microRNA family. Cell 120: 635–647.
Jopling, C.L., Yi, M., Lancaster, A.M., Lemon, S.M., Sarnow, P. 2005. Modulation of hepatitis C virus RNA abundance by a liver-specific MicroRNA. Science 309:1577-81.
Kloosterman, W.P., Wienholds, E., de Bruijn, E., Kauppinen, S., Plasterk, R.H. 2006. In situ detection of miRNAs in animal embryos using LNA-modified oligonucleotide probes. Nat Methods. 3: 27-9.
Koshkin, A.A., Singh, S.K., Nielsen, P., Rajwanshi, V.K., Kumar, R., Meldgaard, M., Ol-sen, C.E. and Wengel, J. 1998. LNA (Locked Nucleic Acids): Synthesis of the adenine, cytosine, guanine, 5-methylcytosine, thymine and uracil bicyclonucleoside monomers, oligomerisation and unprecedented nucleic acid recognition. Tetrahedron 54: 3607-3630.
Krek, A., Grun, D., Poy, M.N., Wolf, R., Rosenberg, L., Epstein, E.J., MacMenamin, P., da Piedade, I., Gunsalus, K.C., Stoffel, M. and Rajewsky, N. 2005. Combinatorial microRNA target predictions. Nat. Genet. 37: 495–500.
Krützfeldt, J., Rajewsky, N., Braich, R., Rajeev, K.G., Tuschl, T., Manoharan, M. and Stoffel, M. 2005. Silencing of microRNAs in vivo with ‘antagomirs’. Nature 438: 685-9.
Kurreck, J., Wyszko, E., Gillen, C. and Erdmann, V.A. 2002. Design of antisense oligonucleotides sta-bilized by locked nucleic acids. Nucleic Acids Res. 30: 1911-1918.
Leaman, D., Chen, P,Y., Fak, J., Yalcin, A., Pearce, M., Unnerstall, U., Marks, D.S., Sander, C., Tuschl, T., Gaul, U. 2005. Antisense-mediated depletion reveals essential and specific functions of microRNAs in Drosophila development. Cell 121: 1097-108.
Lecellier, C.H., Dunoyer, P., Arar, K., Lehmann-Che, J., Eyquem, S., Himber, C., Saib, A., Voinnet, O. 2005. A cellular microRNA mediates antiviral defense in human cells. Science 308: 557-60.
Lewis, B.P., Burge, C.B., Bartel, D.P. 2005. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 120: 15-20.
Lu. J., Getz, G., Miska, E.A., Alvarez-Saavedra, E.A., Lamb, J., Peck, D., Sweet-Cordero, A., Ebert, B.L., Mak, R.H., Ferrando, A.A., Downing, J.R., Jacks, T., Horvitz, H.R.,and Golub, T.R. 2005. Mi-croRNA expression profiles classify human cancers. Nature 435: 834–838.
Michael, M.Z., SM, O.C., van Holst Pellekaan, N.G., Young, G.P. and James, R.J. 2003. Reduced ac-cumulation of specific microRNAs in colorectal neoplasia. Mol. Cancer Res. 1: 882– 891.
Válóczi, A., Hornyik, C., Varga, N., Burgyán, J., Kauppinen?, S. and Havelda, Z. 2004. Sensitive and specific detection of microRNAs by northern blot analysis using LNA-modified oligonucleotide probes. Nucleic Acids Res. 32: e175.
Wahlestedt, C., Salmi, P., Good, L., Kela, J., Johnsson, T., Hokfelt, T., Broberger, C., Por-reca, F., Lai, J., Ren, K., Ossipov, M., Koshkin, A., Jacobsen, N., Skouv, J., Oerum, H., Jakobsen, M.H. and Wengel, J. 2000. Potent and nontoxic antisense oligonucleotides containing locked nucleic acids. Proc.Natl.Acad.Sci.U.S.A. 97: 5633-5638.
Wienholds, E., Kloosterman, W.P., Miska, E., Alvarez-Saavedra, E., Berezikov, E., de Bruijn, E., Hor-vitz, H.R., Kauppinen, S. and Plasterk, R.H.A. 2005. MicroRNA expression in zebrafish embryonic de-velopment. Science 309: 310-311.
Xie, X., Lu, J., Kulbokas, E.J., Golub, T.R., Mootha, V., Lindblad-Toh, K., Lander, E.S. and Kellis, M. 2005. Systematic discovery of regulatory motifs in human promoters and 30 UTRs by comparison of several mammals. Nature 434: 338–345.
Ørom, U.A., Kauppinen, S. and Lund, A. 2006. LNA-modified oligonucleotides mediate specific inhibi-tion of microRNA function, Gene 372: 137-141.




