New GLP-1 therapy Foundayo is Eli Lilly’s second obesity medicine to be authorised for use in the US.
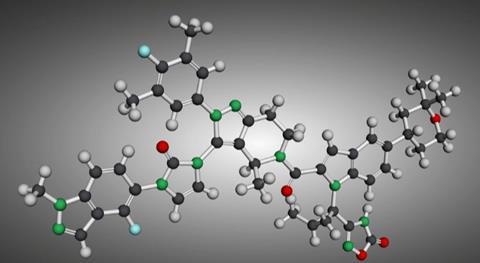
The US Food and Drug Administration (FDA) has approved Eli Lilly’s small molecule Foundayo (orforglipron) for obesity, offering a new option with potential to shift an increasingly competitive market.
A year ago, Lilly published its latest clinical data on the oral glucagon-like peptide-1 (GLP-1) receptor agonist in obesity, and for those who are overweight with weight-related medical problems.
The FDA’s decision means the drug is indicated alongside a reduced-calorie diet and increased physical activity. It can be taken any time of day, with or with food or water.
Writing on LinkedIn, Steve Harvey, CEO of Camena Bio, expects Foundayo’s approval will be “disruptive to the [obesity] market”, due to its novel form. As a small molecule drug, it is “stable in stomach acid”, meaning no injections or restrictions to water intake are required.
Deborah Horn, Director of Obesity Medicine at UTHealth Houston said: “People living with obesity need treatment options that meet them where they are – and for many, a once-daily pill that can be taken with no food or water restrictions can offer them greater flexibility in how they approach their treatment.”
People living with obesity need treatment options that meet them where they are – and for many, a once-daily pill that can be taken with no food or water restrictions can offer them greater flexibility in how they approach their treatment”
Having transitioned through “FDA filing to approval in 50 days”, Harvey noted, Foundayo will now compete with other major players, including Novo Nordisk’s Wegovy (semaglutide), set to lose patent protection in 2028.
The FDA’s approval is based on two phase III trials, ATTAIN-1 and ATTAIN-2. Findings from ATTAIN-1 showed that Foundayo enabled participants to lose an average of 27 pounds, or “an average of 12.4 percent weight loss” at the highest dose in clinical trials, Horn noted.
Lilly’s Chair and CEO, David Ricks, remarked that Foundayo’s approval increases patient access to GLP-1 therapies, and could ultimately “help level the playing field for those living with obesity or who are overweight and living with weight-related complications”.
The firm plans to produce the small molecule GLP-1 therapy at a new manufacturing facility in Alabama, US. At the time of the announcement, Ricks stated this will help Lilly continue its ”onshoring of active pharmaceutical ingredient (API) production, strengthening supply chain resilience and reliable access to medicines for patients in the US.”















No comments yet