The MAS-100 Libra® is an automated passive air sampling system for cleanrooms, combining reliable microbiological monitoring with preserved laminar air flow, while supporting continuous air monitoring in line with the Annex 1 (EudraLex 2022) requirement for critical areas.
Automated passive air monitoring in cleanroom environments
Microbiological air monitoring is a critical component of contamination control in pharmaceutical manufacturing, biotechnology and cleanroom laboratories. Passive air sampling using settle plates is a well-established method, but manual handling can introduce variability and operator-dependent errors. The MAS-100 Libra was developed to automate passive air sampling while maintaining full compliance with cleanroom air flow requirements. By automating the exposure and positioning of agar plates, the system improves reproducibility, reduces manual intervention and supports standardised environmental monitoring programmes.
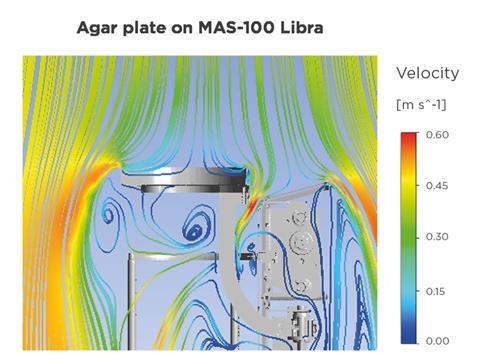
A key concern when introducing any device into a cleanroom is its potential impact on laminar air flow. Even small disturbances can affect particle and microorganism deposition, leading to non-representative results. Computational fluid dynamics (CFD) testing demonstrates that the air flow patterns between a standard agar plate setup – representing a traditional settle plate in a simple holder – and the MAS-100 Libra are comparable.
Objective of the air flow analysis
The CFD study aimed to determine whether the MAS-100 Libra alters laminar air flow in a way that could compromise passive air sampling results. In line with EU GMP Annex 1, cleanroom air flow is typically unidirectional at ~0.45 m/s. Any turbulence, recirculation or backflow near the sampling surface could bias microbial recovery. Therefore, the air flow behaviour around the exposed agar plate within the MAS-100 Libra was directly compared with that around a standalone settle plate.
CFD simulation methodology
The simulation model represented a simplified cleanroom with vertical laminar air flow. Two configurations were analysed: a single exposed agar plate and the MAS-100 Libra with an exposed plate positioned on its sampling surface. Velocity vectors, streamlines and turbulence intensity were evaluated at different heights and cross-sections above and below the sampling area. This approach allowed a detailed comparison of air flow conditions relevant to microbial deposition on the agar surface.
Key results and observations
CFD results demonstrated that air flow directly above the agar plate in the MAS-100 Libra closely matched the air flow observed above a free settle plate. This indicates that the device does not significantly interfere with the ‘first air’ reaching the sampling surface. As a result, microorganisms carried by the cleanroom air flow can deposit on the agar plate in a representative manner, preserving the integrity of passive air sampling.
Increased turbulence was observed below the sampling platform and around the housing of the instrument. However, these effects occurred outside the critical sampling zone and did not influence the air flow above the agar plate.
Importantly, no backflow from the instrument onto the agar surface was detected. This confirms that air is not redirected from the device onto the sampling area, eliminating the risk of biased contamination results caused by disturbed air flow patterns.
Relevance for cleanroom monitoring and decontamination
The CFD analysis revealed that air flow streamlines enter the internal structure of the instrument. This finding is particularly relevant for vaporised hydrogen peroxide (VHP) decontamination processes, as it indicates that the internal components of the instrument are exposed to the decontamination agent. This supports the validation of cleaning and sterilisation procedures in high-grade cleanroom applications.
Conclusion
The MAS-100 Libra provides a validated, automated solution for passive air sampling in cleanrooms without compromising laminar air flow. It ensures representative microbial sampling, improves consistency, reduces operator influence and enhances process control. This makes the MAS-100 Libra a valuable instrument for pharmaceutical and cleanroom environments with stringent contamination control requirements.
For further information, visit: mbv.ch















No comments yet