RNAi therapeutics: addressing targets?
Posted: 2 August 2008 | Dr Mark Edbrooke and Dr Neil Clarke, GlaxoSmithKline Research and Development, Hertfordshire | No comments yet
Gene silencing by RNA interference (RNAi) uses double-stranded RNA to shut down gene expression in cells. This provides the possibility that this new methodology could be used in the treatment of disease symptoms and disease processes. A particular attraction of RNAi (as well as other gene knockdown methods of treatment, including antisense) is that, at present, no one class of protein appears to be refractive to silencing using this method and, therefore, it has the potential to make any gene product a target for pharmaceutical intervention. As will be discussed later though, delivery of these large polyanionic molecules to their site of action may pose a significant challenge.
Gene silencing by RNA interference (RNAi) uses double-stranded RNA to shut down gene expression in cells. This provides the possibility that this new methodology could be used in the treatment of disease symptoms and disease processes. A particular attraction of RNAi (as well as other gene knockdown methods of treatment, including antisense) is that, at present, no one class of protein appears to be refractive to silencing using this method and, therefore, it has the potential to make any gene product a target for pharmaceutical intervention. As will be discussed later though, delivery of these large polyanionic molecules to their site of action may pose a significant challenge.
Gene silencing by RNA interference (RNAi) uses double-stranded RNA to shut down gene expression in cells. This provides the possibility that this new methodology could be used in the treatment of disease symptoms and disease processes. A particular attraction of RNAi (as well as other gene knockdown methods of treatment, including antisense) is that, at present, no one class of protein appears to be refractive to silencing using this method and, therefore, it has the potential to make any gene product a target for pharmaceutical intervention. As will be discussed later though, delivery of these large polyanionic molecules to their site of action may pose a significant challenge.
RNA interference (RNAi)
Professors Andrew Fire and Craig Mello were awarded the 2006 Nobel Prize in Medicine for their work on RNA interference in the nematode worm, Caenorhabditis elegans1. However, this work involved the use of long double-stranded RNA molecules to shut down gene expression, and it was already known at that time that RNA of this size leads to interferon-mediated cell death if introduced into mammalian cells2. The discovery by Sayda Elbashir and colleagues in 2001, that short duplexes of RNA could mediate RNAi in mammalian cells without triggering interferon responses3, has spawned a whole new world of functional genomics and, ultimately, the possibility of new treatments for disease.
The mechanism by which RNAi works has subsequently been characterised in much detail4. The double-stranded RNA molecule, once inside the cell, is unwound in the cytoplasm to release the two strands of RNA. The sense or ‘passenger’ strand is degraded, but the antisense or ‘guide’ strand becomes incorporated into a silencing complex known as RISC (RNA-induced silencing complex). This complex then tracks down messenger RNAs (mRNAs) that are complimentary in sequence to the ‘guide’ strand, and an exonuclease enzyme in the complex known as Dicer cleaves and destroys the mRNA. The complex is then available to pursue further mRNA molecules for destruction.
The double-stranded RNA molecules are usually 19 to 23 base pairs in length, and can be synthesised on an oligonucleotide synthesiser. These molecules are known as small interfering RNAs, or siRNAs. They can also be generated as hairpin loop structures using a vector system, for example from a plasmid or a virus, and are known as short hairpin RNAs (shRNAs)5,6. The Dicer enzyme is then involved in processing these molecules into structures that are very similar to siRNAs, which are then incorporated into RISC in a similar fashion to siRNAs oligonucleotides.
Advantages and challenges of RNAi
A major advantage of RNAi is the generic mechanism that results in sequence-specific mRNA targeting. The effector molecules utilise a common catalytic mechanism that provides high potency and low toxicity, with broad and predictable application. The generic nature of the mechanism has the potential to unlock many validated disease targets that have been ‘undruggable’ to date. siRNAs and/or shRNAs can be used in combination in order to inhibit multiple targets at the same time, or to decrease the possibility of mutational escape by infectious agents. With regards to therapeutic development, RNAi effector molecules can be designed and validated in a matter of months, leading to the potential for significantly decreased cycle time. Also, the oligonucleotides can be modified or conjugated in order to reduce the possibility of degradation, to prevent off-target effects from the ‘passenger’ strand, and to enhance their tissue- and cell-penetrating abilities without any loss of potency7 (see Figure 1).
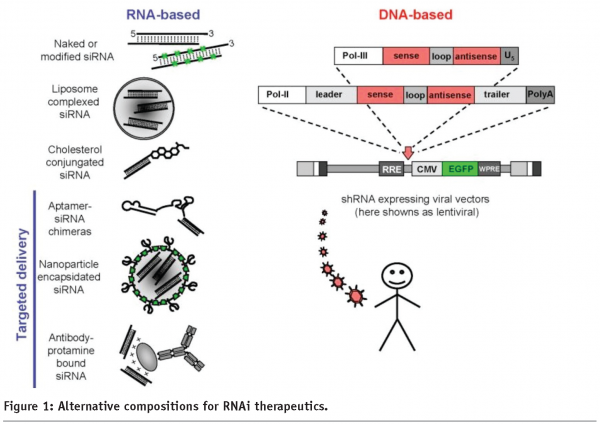

Product delivery is undoubtedly the biggest challenge in the development of RNAi therapeutics. Unlike small molecule drugs, RNAi effector molecules are relatively large and polyanionic in nature. This impacts their ability to gain access to the tissue of interest and then gain entry into the cytoplasm of the target cells. Delivery has to be achieved with minimal loss or degradation whilst in transport to the tissue, but with suitable release of the effector molecules once inside the cells. The delivery challenge means that the first RNAi drug developed is likely to be restricted to the most accessible organs. However, it is expected that this range of organs will increase over time with the intensive effort going into developing a variety of new delivery technologies. Also, despite the exquisite sequence specific nature of RNAi effector molecules, there is the possibility that they can hybridise to, and therefore interfere with, inappropriate mRNAs. It should also be born in mind that RNAi leads to the knockdown of target gene expression i.e. it works as an antagonist. Therefore, if agonism of the target is what is required to achieve a therapeutic benefit then an alternative form of drug may be required. Finally, cost-of-goods is currently still an issue as oligonucleotide synthesis is not as cheap as small molecule synthesis. However, these costs are steadily being reduced as synthesis efficiencies are being made. The same financial considerations have to be applied if the RNA molecules are to be formulated or encapsulated in a multi-component complex in order to facilitate cellular delivery.
The current clinical pipeline for RNAi
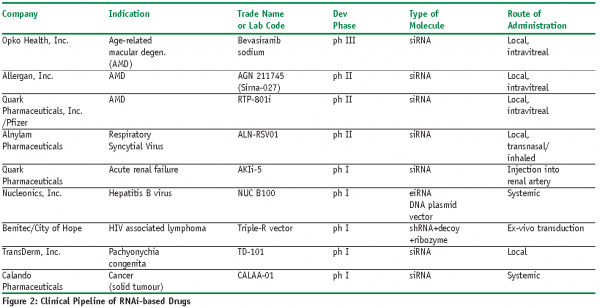
There are currently nine ongoing clinical trials using siRNA oligonucleotides or shRNA vectors. The majority of these are using local or ‘topical’ delivery in order to avoid the challenges of systemic delivery of these large molecules. Opko Health, Inc. is the most advanced with an siRNA-based treatment for age-related macular degeneration (AMD)8. The siRNA targets VEGF production, and is delivered by intravitreal injection on a six to eight weekly basis. This asset is now in Phase III clinical trials having shown significant efficacy in wet AMD patients in phase II trials. Recent data has also shown that ‘bevasiranib’, Opko’s siRNA molecule, is distributed throughout the eye, including extensive uptake into the retina9. Interestingly, it is being evaluated as a co-treatment with Lucentis, a monoclonal antibody also targeting VEGF. Note that the oligonucleotides are unmodified as the eye has only very low levels of RNA degrading enzymes. Two other companies are also developing treatments for AMD including Allergan, Inc. and Quark Pharmaceuticals, Inc./Pfizer, although the Quark compound is claimed to be a ‘modified siRNA’10,11.
Another topically delivered siRNA is Alnylam Pharmaceutical’s anti-respiratory syncytial virus (RSV) candidate that has recently shown clinical proof of principle12. This uses a simple formulation of naked siRNA, and has been delivered intranasally. Alnylam are now planning more complex inhaled delivery for this candidate. It should be noted that topically delivered siRNAs against RSV and parainfluenza virus (PIV) have already been shown to reduce the in vivo viral load of these two viruses in animals by greater than three orders of magnitude13. Similarly, siRNA formulated in 5% dextrose in water (D5W) administered intranasally to a non-human primate model of SARS corona virus infection significantly reduced interstitial infiltrates and pathological changes14.
Quark Pharmaceuticals have started Phase I trials with a chemically modified siRNA for an undisclosed target for acute renal failure (ARF, also known as acute kidney injury, AKI)15. The siRNA acts to temporarily and reversibly inhibit the expression of human p53. This treatment is being delivered by injection and could therefore be construed as being ‘systemic’. However, delivery is directly into the renal artery, so is more akin to a topical delivery route.
Nucleonics, Inc. is using a plasmid vector for systemic delivery of shRNAs (known as expressed interfering RNAs, eiRNA) to the liver for the treatment of hepatitis B virus (Hep B)16. This vector produces four different discrete siRNA-like molecules, all targeting the Hep B virus, with the intention that the multiple targeting will reduce or hopefully prevent the generation of ‘drug escape mutants’ by the virus. Benitec/City of Hope are using a lentiviral vector that expresses a shRNA as well as a decoy RNA and a ribozyme to target HIV infection17. The shRNA and the decoy both target specific proteins from the virus (tat/rev and TAR respectively), and the ribozyme aims to silence the synthesis of a cellular host receptor for the virus, CCR5. Stem cells are harvested from HIV-infected patients, treated with the lentivirus, and then reinfused into the patients during a bone marrow transplant. In a similar fashion to the Nucleonics therapy, it is hoped that addressing multiple targets using three different modes of inhibition will significantly reduce the likelihood that the virus will be able to escape the treatment.
TransDerm, Inc. are using topical delivery to treat pachyonychia congenita, and are targeting the N171K mutation in keratin 6a (K6a)18. Pachyonychia congenita is a rare, hereditary, autosomal dominant palmoplantar keratoderma which is caused by mutations in genes for keratin K6a, K6b, K16 and K17, its most common and prominent features include hypertrophic nail dystrophy, focal palmar and plantar hyperkeratosis (amongst others). Again, this is ‘local’ delivery of unmodified siRNAs, though TransDerm are also developing a product called GeneCreme which will allow the siRNA to be topically applied to the skin in a patient-friendly manner.
Finally, Calando Pharmaceuticals have recently commenced Phase I studies using a cyclodextrin-based polymer nanoparticle to deliver a siRNA that targets ribonucleotide reductase, a treatment for solid tumours19.
The delivery challenge
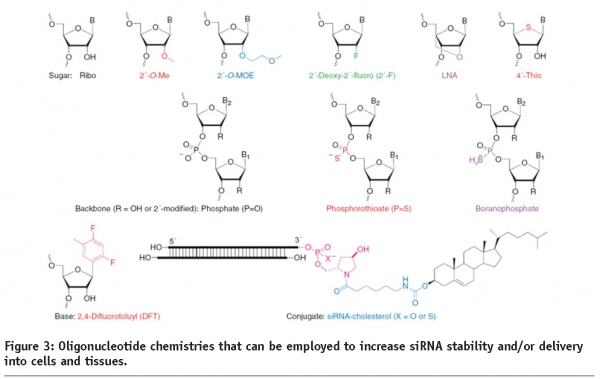
For RNAi therpeutics to realise their full potential, the challenge of tissue and cellular delivery have to be overcome. There are numerous potential routes to get past this challenge, and companies and academic groups are trying multiple different approaches. To help achieve successful delivery, the drug molecule can be chemically modified to prevent degradation and also to enhance binding to plasma proteins. A typical modification is the replacement of non-bridging oxygen molecules with sulphur groups in the backbone of the oligonucleotide to generate ‘phosphorothioate’ molecules. Other modifications include additions to the 2’ position of the ribose in order to reduce degradation, to avoid immune stimulation, and also to prevent unwanted ‘off target’ binding by the sense strand of the siRNAs (see references 20 and 21 for a summary). Alnylam Pharmaceuticals, Sirna Therapeutics, and Silence Therapeutics are all using varying degrees of chemical modification in their oligonucleotides for their pre-clinical trials (see Figure 2).

Oligonucleotides can also be coupled to a second component that helps direct them to specific tissues. For example, cholesterol has been used to guide these molecules to the liver22. Also, the use of aptamers23,24 and monoclonal antibodies (mAbs), Fab, and scFv fragments have been investigated25.
In particular for systematic delivery, companies are combining the oligonucleotides with small lipid, polymer or other types of transporter molecules, termed ‘nanoparticles’. These increase their residence time in tissues and also facilitate their entry into the target cell. An example of lipid encapsulation is the approach taken by Alnylam Pharmaceuticals where they have used SNALPs (stable nucleic acid lipid particles, developed by Protiva) in order to achieve significant delivery to the liver after intravenous injection into mice. This method was shown to be highly successful for the delivery of siRNAs targeting apolipoprotein B (apoB) in cynomolgus monkeys26. Other companies and academic groups are also developing lipid-, peptide-, or polymer-based nanoparticles. Novosom AG have developed a charge-reversible liposomal technology known as SMARTICLES for the delivery of RNA- and DNA-based therapeutics27, and RXi Pharmaceuticals intend to exploit the use of proprietary nanotransporters in collaboration with the laboratory of Tariq Rana, University of Massachusetts Medical School28.
MDRNA, Inc. (formerly known as Nastech Pharmaceutical Co. Inc.) are exploring the use of both lipid- and peptide-based complexes, and Mirus Bio Corporation are using ‘dynamic’ polyconjugates that employ pH-dependent carboxy dimethylmaleic anhydride chemistry to release siRNAs when inside cells29. Calando Pharmaceuticals and Intradigm Corporation are both developing polymer-based nanoparticles19,30. The large number of companies that are currently involved in developing delivery methodologies for RNA-based therapeutics is a direct reflection of the challenges that are faced by trying to successfully deliver large polyanionic molecules into the variety of tissues and cells in the body.
Non-topical delivery to specific tissues and organs is another whole world of delivery challenge, which will be covered in a future article in this magazine. In the meantime, the recent success of Kumar et al., to achieve delivery to the CNS using a short peptide derived from rabies virus glycoprotein shows that delivery to any organ may, ultimately, be possible31.
Finally, other components involved in the successful delivery of RNA-based therapeutics must not be overlooked including RNAi patches, creams and/or inhalers.

Summary
To a major degree, the initial set of RNAi-based clinical trials are using local delivery, and are addressing targets that have already been shown to be druggable by other means e.g. small molecules and/or monoclonal antibodies. The next stage in the development of RNAi as an effective therapeutic will be to achieve successful delivery of these molecules to more difficult cells and tissues. If this can be achieved, then addressing targets than have previously been deemed to be ‘undruggable’ by traditional methods is the next big step.Ultimately, this may well lead to the treatment of new disease targets and previously untreated diseases.
References
- Fire, A., Xu, S., Montgomery, M.K., Kostas, S.A., Driver, S.E., and Mello, C.C. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 1998, 391(6669):806-811.
- Stark, G.R, Kerr, I.M., Williams, B.R., Silverman, R.H. and Schreiber, R.D. How cells respond to interferons. Annu. Rev. Biochem. 1998, 67:227-264.
- Elbashir, S.M., Harborth, J., Lendeckel, W., Yalcin, A., Weber, K. and Tuschl, T. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 2001, 411:494-498.
- Kim, D.H. and Rossi, J.J. Strategies for silencing human disease using RNA interference. Nature Rev. Genet. 2007, 8(3):173-184.
- Brummelkamp, T.R., Bernards, R. and Agami, R. A system for stable expression of short interfering RNA in mammalian cells. Science 2002, 296:550-553.
- Paddison, P.J., Caudy, A.A., Bernstein, E., Hannon, G.J. and Conklin, D.S. Short hairpin shRNAs induce sequence-specific silencing in mammalian cells. Genes Dev. 2002, 16(8):948-958.
- de Fougerolles, A., Vornlocher, H-P., Maraganore, J. and Lieberman, J. Interfering with disease: a progress report on siRNA-based therapeutics. Nat. Rev. Drug Discov. 2007, 6(6):443-453.
- OPKO Health Initiates Phase 3 Trial of Bevasiranib for the Treatment of AMD. Trial Designed to Compare Efficacy of Bevasiranib Administered Every 8 Weeks or 12 Weeks with Lucentis® Administered Every 4 Weeks. Press Release 11th July 2007.
- Dejneka, N.S., Wan, S., Bond, O.S., Kornbrust, D.J. and Reich, S.J. Ocular biodistribution of bevasiranib following a single intravitreal injection to rabbit eyes. Molecular Vision 2008, 14:997-1005.
- NCT00395057: A Study Using Intravitreal Injections of a Small Interfering RNA in Patients With Age-Related Macular Degeneration. http://clinicaltrials.gov/ct2/show/NCT00395057
- The proposed primary indication for RTP801i-14 is the treatment of patients with neovascular (wet) Age-related Macular Degeneration (AMD or ARMD). Public information on Quark web site. http://www.quarkpharma.com/qbi-en/products/53/
- Alnylam Achieves First Human Proof of Concept for an RNAi Therapeutic with GEMINI Study. Press Release 29th February 2008. http://phoenix.corporate-ir.net/phoenix.zhtml?c=148005&p=irol-newsArticle&ID=1113937&highlight=
- Bitko, V., Musiyenko, A., Shulyayeva, O. and Barik, S. Inhibition of respiratory viruses by nasally administered siRNA. Nature Med. 2005, 11:50-55.
- Li, B.-j., Tang, Q., Cheng, D., Qin, C., Xie, F.Y., Wei, Q., Xu, J., Liu, Y., Zheng, B.-j., Woodle, M.C., Zhong, N. and Lu, P.Y. Using siRNA in prophylactic and therapeutic regimens against SARS coronavirus in Rhesus macaque. Nature Med. 2005, 11:944-951.
- Quark Pharmaceuticals, Inc. Presented Positive Preclinical Results of Systemic RNAi Compound for Acute Renal Failure (ARF). Quark to initiate Phase 1 trial of AKIi-5 as the first human clinical study involving the systemic delivery of siRNA. Press Release 2nd August 2007.
- Romano, P.R., McCallus, D.E., and Pachuk, C.J. RNA interference-mediated prevention and therapy for hepatocellular carcinoma. Oncogene 2006, 25:3857-3865.
- Anderson, J., Li, M-J., Palmer, B., Remling, L., Li, S., Yam, P., Yee, J.-K., Rossi, J., Zaia, J. and Akkina, R. Safety and efficacy of a lentiviral vector containing three anti-HIV genes – CCR5 ribozyme, Tat-rev siRNA, and TAR decoy – in SCID-hu mouse-derived T cells. Molecular Therapy 2007, 15(6):1182-1188.
- Smith, F.J.D., Hickerson, R.P., Sayers, J.M., Reeves, R.E., Contag, C.H., Leake, D., Kaspar, R.L. and McLean, W.H.I. Development of Therapeutic SiRNAs for Pachyonychia Congenita. J. Invest. Dermatol. 2007, 128:50-58.
- Heidel, J.D., Yu, Z., Liu, J.Y.-C., Rele, S.M., Liang, Y., Zeidan, R.K., Kornbrust, D.J. and Davis, M.E. Administration in non-human primates of escalating intravenous doses of targeted nanoparticles containing ribonucleotide reductase subunit M2 siRNA. Proc. Natl. Acad. Sci. U.S.A. 2007, 104:5715-5721.
- Bumcrot, D., Manoharan, M., Koteliansky, V. and Sah, D.W.Y. RNAi therapeutics: a new class of pharmaceutical drugs. Nature Chem. Biol. 2006, 2(12):711-719.
- Judge, A.D., Bola, G., Lee, A.C.H. and MacLachlan, I. Design of Noninflammatory synthetic siRNA Mediating Potent Gene Silencing in Vivo. Molecular Therapy 2006, 13(3):494-505.
- Soutschek, J., Akinc, A., Bramlage, B., Charisse, K., Constien, R., Donoghue, M., Elbashir, S., Geick, A., Hadwiger, P., Harborth, J., John, M., Kesavan, V., Lavine, G., Pandey, R.K., Racie, T., Rajeev, K.G., Rohl, I., Toudjarska, I., Wang, G., Wuschko, S., Bumcrot, D., Koteliansky, V., Limmer, S., Manoharan, M. and Vornlocher, H.-P. Therapeutic silencing of an endogenous gene by systemic administration of modified siRNAs. Nature 2004, 432:173-178.
- McNamara, J.O., Andrechek, E.R., Wang, Y., Viles, K.D., Rempel, R.E., Gilboa, E., Sullenger, B.A. and Giangrande, P.H. Cell type-specific delivery of siRNAs with aptamer-siRNA chimeras. Nature Biotech. 2006, 24:1005-1015.
- Zhou, J., Li, H., Li, S., Zaia, J. and Rossi, J.J. Novel dual inhibitory function aptamer-siRNA delivery system for HIV-1 therapy. Mol Therapy 2008, (06 May 2008) available on line.
- Song, E., Zhu, P., Lee, S.-K., Chowdhury, D., Kussman, S., Dykxhoorn, D.M., Feng, Y., Palliser, D., Weiner, D.B., Shankar, P., Marasco, W.A. and Lieberman, J. Antibody mediated in vivo delivery of small interfering RNAs via cell-surface receptors. Nature Biotech. 2005, 23(6):709-717.
- Zimmermann, T.S., Lee, A.C.H., Akinc, A., Bramlage, B., Bumcrot, D., Fedoruk, M.N., Harborth, J., Heyes, J.A., Jeffs, L.B., John, M., Judge, A.D., Lam, K., McClintock, K., Nechev, L.V., Palmer, L.R., Racie, T., Rohl, I., Seiffert, S., Shanmugam, S., Sood, V., Soutschek, J., Toudjarska, I., Wheat, A.J., Yaworski, E., Zedalis, W., Koteliansky, V., Manoharan, M., Vornlocher, H.-P. and MacLachlan, I. RNAi-mediated gene silencing in non-human primates. Nature 2006, 441:111-114.
- SMARTICLES® define a novel class of liposomes: fully charge-reversible particles. Public information on Novosom web site. http://www.novosom.com/smart_01.asp
- Baigude, H., McCarroll, J., Yang, C.-s., Swain, P.M. and Rana, T.M. Design and creation of new nanomaterials for therapeutic RNAi. ACS Chem Biol. 2007, 2(4):237-241.
- Rozema, D.B., Lewis, D.L., Wakefield, D.H., Wong, S.C., Klein, J.J., Roesch, P.L., Bertin, S.L., Reppen, T.W., Chu, Q., Blokhin, A.V., Hagstrom, J.E., and Wolff, J.A. Dynamic polyconjugates for targeted in vivo delivery of siRNA to hepatocytes. Proc. Natl. Acad. Sci. U.S.A. 2007, 104:12982-12987.
- Intradigm’s RNAi Nanoplex (NPX) Delivery Technology. Public information on Intradigm web site. http://www.intradigm.com/technology.html
- Kumar, P., Wu, H., McBride, J., Jung, K.-E., Kim, M.H., Davidson, B.L., Lee, K.S., Shankar, P. and Manjunath, N. Transvascular delivery of small interfering RNA to the central nervous system. Nature 2007, 448:39-43.






