A new era for microcalorimetry in drug development
Posted: 21 September 2007 | | No comments yet
Drug development involves the identification and subsequent optimisation of low molecular weight compounds with a desired biological activity. Often, the initial binding affinity of those compounds towards their intended target needs to be improved by five or more orders of magnitude before they become viable drug candidates; a process that would be greatly facilitated if the different forces that contribute to binding were experimentally accessible. Isothermal titration calorimetry (ITC) provides such a tool.
Drug development involves the identification and subsequent optimisation of low molecular weight compounds with a desired biological activity. Often, the initial binding affinity of those compounds towards their intended target needs to be improved by five or more orders of magnitude before they become viable drug candidates; a process that would be greatly facilitated if the different forces that contribute to binding were experimentally accessible. Isothermal titration calorimetry (ITC) provides such a tool.
Drug development involves the identification and subsequent optimisation of low molecular weight compounds with a desired biological activity. Often, the initial binding affinity of those compounds towards their intended target needs to be improved by five or more orders of magnitude before they become viable drug candidates; a process that would be greatly facilitated if the different forces that contribute to binding were experimentally accessible. Isothermal titration calorimetry (ITC) provides such a tool.
Of all the techniques available to measure binding, ITC is the only one capable of measuring not only binding affinities, but also the different thermodynamic forces that determine the binding energy. In the past, however, ITC has been used retrospectively rather than as a guiding tool for lead optimisation. This situation is changing due to two factors: improved understanding of the relationships between thermodynamic forces (enthalpy, entropy and heat capacity) and chemical structure, and a new generation of instruments with reduced sample requirements and much faster throughputs. In this article these developments, which anticipate a new era in microcalorimetry, will be discussed.
Prevalent strategies in drug discovery rely heavily on the screening of large libraries of compounds. Most often, targets for drug development are enzymes and the screening is performed by implementing enzyme inhibition assays in a high throughput format that allows the rapid identification of compounds with inhibitory activity. Usually, compounds identified by screening ‘hits’ have binding affinities in the low to high micromolar range, requiring potency improvements of five or more orders of magnitude before they can become viable drug candidates. Nevertheless, these hits provide the starting material for further development.
Once hits are identified, important questions need to be answered ranging from areas such as bioavailability, toxicity and pharmacokinetics, to potency and selectivity. Decisions also need to be made in order to select the most appropriate scaffolds for further development. In the area of potency and selectivity (the subject of this article) important questions include: Which scaffolds are the most appropriate for further optimisation? Which scaffolds have the potential to be optimised? What is the best strategy for potency optimisation? What is the best strategy for selectivity optimisation? The correct answers to those questions are of the upmost importance as the wrong decision might lead to a dead end rather than a successful drug.
The forces of binding
Different forces determine binding affinity. These forces can be classified as attractive forces between the ligand and target molecules of which Van der Waals and hydrogen bonds are the most prominent, and repulsive forces between the drug and solvent of which the hydrophobic effect is the most important. The strength of Van der Waals interactions depends on how closely the ligand molecule fits within the binding pocket (shape complementarity), while the strength of hydrogen bonds depends on the distance and angle between hydrogen bond acceptors and donors in the ligand and target molecules. In fact, these constraints are so strict that weak hydrogen bonds often end up opposing rather than favouring binding, as desolvation penalties and compensatory entropic effects predominate. The hydrophobic effect, on the other hand, does not represent an attraction between ligand and target but an exclusion of the ligand from the solvent.
It is evident that while all these forces contribute to potency, not all of them contribute equally to selectivity. A tight fit between ligand and binding pocket not only maximises Van der Waals interactions but also lowers the probability that the ligand will accommodate equally well in other protein. Hydrogen bonds are also major contributors to selectivity due to their stringent distance and angle constraints. A different arrangement of donors and acceptors within the binding pocket not only weakens or eliminates the favourable energy of hydrogen bonds, but leaves intact the severe desolvation penalty that originates from the burial of unsatisfied polar groups, thus lowering the affinity towards unwanted targets. Hydrophobicity alone, does not contribute to selectivity as it is not an attractive force to the target.
For binding to occur, the favorable forces mentioned above need to overcome the unfavorable entropy associated with restricting translational/rotational and conformational degrees of freedom of ligand and protein. Of these entropic terms, the conformational entropy of the ligand molecule is the only one that can be readily manipulated by the introduction of conformational constraints that approximate the free ligand conformation to its bound conformation. In this fashion, the conformational entropy loss of the ligand is minimised.
There are basically four binding forces that can be improved by the drug designer in order to optimise the affinity and selectivity of a ligand:
- Better Van der Waals interactions by improving the geometrical fit of the ligand within the binding pocket
- The formation of strong hydrogen bonds
- Increased hydrophobicity
- Minimisation of conformational entropy losses by conformationally constraining and pre-shaping the ligand molecule to the target.
The outcome of screening
High throughput screening generally produces hits with weak binding affinities. Binding affinities in the micromolar range correspond to Gibbs energies of binding of -5 to -8 kcal/mol. This binding energy level can be achieved by many combinations of forces, particularly combinations in which different forces point in opposite directions. In high throughput screening, the influence of different binding forces in the outcome are not equally weighted and the resulting hits are usually biased towards hydrophobicity. For example, it is highly improbable that a ligand selected at random will closely fit the binding pocket of the target molecule. Hydrogen bonds, on the other hand, require precise distance and angular constraints that are very unlikely to occur randomly. While there is a certain probability for one hydrogen bond, multiple hydrogen bonds are highly unlikely. Moreover, molecules with unsatisfied hydrogen bond donors or acceptors are penalised by a high desolvation penalty and exhibit very weak potency. At this early stage hydrophobicity combined with certain size limitations are sufficient to generate the necessary energy to achieve micromolar affinity levels. The tendency towards higher hydrophobicity and high throughput screening has been statistically documented by Lipinski1.
Once hits are identified the next step is to profile and prioritize them in order to select those that qualify for further optimisation. At this point, the information obtained with traditional binding assays is usually restricted to the potency of the compounds (IC50 or apparent Ki) and their chemical structures. The chemical structure can provide clues about potential toxicity, bioavailability or pharmacokinetic issues as well as chemical modification potential. This information cannot answer some fundamental questions: Why do the leads bind the target? Do they bind because they establish good interactions with the target or because they are excluded from the solvent? It seems apparent that it would be preferable to select a compound that establishes good interactions with the target rather than one that is excluded from the solvent even if has a lower potency. It is at this level that ITC has begun to play a fundamental role, as already demonstrated in the literature2-4.
Thermodynamic signatures
The binding affinity is determined by the Gibbs energy of binding (ΔG), which is itself a function of the enthalpy (DH) and entropy (Δ S) changes (Δ G = Δ H – T Δ S). Different effects contribute to the Gibbs energy of binding; these effects not only include those directly related to the ligand/target interaction, which are critical during drug optimisation, but also indirect effects such as conformational changes of the protein upon binding, or coupled protonation reactions. In the vast majority of situations those terms constitute a constant background since the ligand under optimisation targets the same site and elicits the same inhibitory effect. Exceptions involve the design of inhibitors aimed at modulating an allosteric effect in which conformational changes need to be monitored during optimisation5.
The binding forces that can be manipulated by the drug designer contribute differently to the enthalpy and entropy changes. Van der Waals interactions and hydrogen bonds contribute favourably to the binding enthalpy, while hydrophobicity contributes primarily to a favourable binding entropy. The introduction of conformational constraints aimed at pre-shaping the ligand to the target minimises unfavourable conformational entropy effects. Changes in these forces are reflected in the thermodynamic signature of the compound as it moves along the optimisation path. The thermodynamic signature provides an instantaneous visual representation of the enthalpic and entropic contributions to the binding energy as shown in Figure 1.
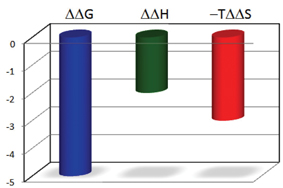

Figure 1: The thermodynamic signature of a compound provides a rapid assessment of the improvement in binding energy (ΔΔ G) and the origin of that improvement (ΔΔ H or -T ΔΔ S). The nature of the improvement permits determination of the required actions for further optimization, for example, better enthalpic or entropic interactions and the necessary compound modifications to bring about those changes. The changes in the thermodynamic parameters are expressed in relation to those of the initial compound in the series, which serves as reference.
The thermodynamic signatures of the initial hits can be used to assess the relative contributions of the different binding forces to their binding affinity. Usually, this is done by selecting one hit (for example the one with the highest potency) and plotting the difference in thermodynamic parameters with the reference (ΔΔ G, ΔΔ H and -T ΔΔ S). Compounds with a larger hydrophobic component will exhibit larger binding entropies while compounds with better van der Waals or hydrogen bonds will exhibit more favourable binding enthalpies. The hit profiling obtained by the thermodynamic signature goes beyond a simple potency ranking since it also provides important insights into the binding mechanisms of the hits identified by screening. This information is critical as frequently a hit can be preferred due to a more robust binding mechanism than simply to the potency exhibited at these early stages.
Once a scaffold has been selected for optimisation, its thermodynamic signature provides a rigorous optimisation blueprint and a way of monitoring progress. This progress monitoring is critical for fast optimisation as the introduction of hydrogen bonds is often compensated by opposing entropic changes resulting in no affinity gains6. In addition, the thermodynamic signature indicates the type of changes, enthalpic or entropic, and the necessary chemistry that is required to improve potency and selectivity.
During optimisation it is common to observe that the enthalpy change becomes unfavorable, especially after the introduction of a polar group. This phenomenon is an indication that the polar group becomes buried and desolvated but does not establish a strong hydrogen bond, the enthalpic contribution being dominated by the unfavorable desolvation enthalpy7. ITC is the only technique that will identify the reasons for the lack of improvement after a specific modification and suggest corrective actions.
Extremely high potency is achieved when both the enthalpy and entropy changes contribute favourably to binding. The evolution of the HIV-1 protease inhibitors provides a clear picture. First, generation protease inhibitors like indinavir are characterised by unfavourable enthalpy changes, while newer inhibitors have strongly favourable enthalpies8-10. A similar situation has been observed for the cholesterol lowering drugs, the statins11.
Isothermal titration calorimetry
Among the techniques used to study binding, ITC is unique, since in just one experiment it is able to determine the Gibbs energy, association constant, enthalpy and entropy changes, as well as the stoichiometry of binding. ITC provides this unique capability in-solution, in a label-free format, and without the need to immobilise either the target macromolecule or ligand12.
In a typical experiment, the ITC measures the heat released, or absorbed by the stepwise addition of a ligand molecule into the reaction cell of the calorimeter containing a solution of the target protein, while maintaining constant temperature and pressure. The change in chemical composition as a result of the addition of ligand, using a precision computer-controlled syringe, triggers the binding reaction. The content of the sample cell is continuously stirred to ensure proper mixing of the reactants. A second cell, where no binding reaction takes place, is used as a reference. The heat released or absorbed after each injection is obtained by calculating the area under each peak. Since the measured heat is proportional to the increase in bound macromolecule concentration, its magnitude decreases as the protein becomes saturated. After saturation, additional injections produce small identical peaks, due to non-specific phenomena such as dilution and mixing effects, whose heat effect must be subtracted from all the injection peaks before performing data analysis. Data analysis of the heat effect for each injection leads to the determination of the thermodynamic binding parameters12,13. The outcome of a typical ITC experiment is shown in Figure 2.
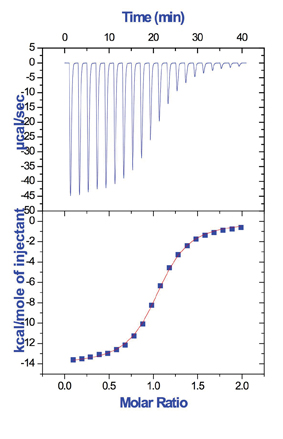

Figure 2: Typical ITC titration of a protein by a small ligand. The signal recorded by the instrument is shown in the top panel. This signal is equal to thermal power required to maintain a constant temperature in the sample cell during the sequence of injections of the ligand into the protein. Depending on the exothermic or endothermic character of the binding reaction, the peaks will show a downward or upward deflection respectively. The area under each peak (integrated heat) for the sequence of injections is shown in the bottom panel. The thermodynamic binding parameters (binding affinity, binding enthalpy and stoichiometry) are obtained through non-linear regression of the experimental data.
New instruments and new applications
While ITC is widely acknowledged as a provider of the gold standard for binding parameters, its widespread utilisation in the early stages of drug development has been limited by two factors: the relatively high amounts of protein required to perform a binding determination (0.1 – 1mg), and the time required to perform the measurement. A new generation of instruments is effectively addressing those issues and creating new applications. For example, the recently released iTC200 from Microcal (Northampton, MA)14 reduces the cell compartment volume by a factor of seven over its predecessor the VP ITC. It does this without a reduction in sensitivity and a significantly faster time response. This innovation permits the performance of experiments with ca. 10 times less protein sample and a total of 2-4 titrations per hour. A smaller cell volume not only facilitates the standard use of ITC but also extends the number of ITC applications. For example, the range of binding affinities that can be measured by ITC is dictated by a parameter called ‘c value’ equal to the product of the binding affinity and the total concentration of macromolecule (c = [Mtotal]Ka)12. For accurate affinity determination the c value must be between 1 and 1000. A decrease in the cell volume by a factor of ten results in a similar increase in c value if the same amount of protein is used, and consequently the ability to measure weak binders. This ability could be important in the early stages of drug discovery, in which binding affinities are weak, especially in conjunction with a fully automated instrument. In this respect, the automated version of the new iTC200 is expected to provide a sample through-put of at least 50 samples per day with a capacity to process as many as 384 samples in an unattended run14.
Summary
The binding affinity is determined by enthalpic and entropic forces. In the past, structure activity relationships (SAR) aimed at improving the affinity of potential drug candidates have been hampered by the absence of information regarding the origin of specific advantageous or detrimental effects, and consequently the absence of clear guidelines for optimisation. New developments relate specific ligand/target interactions and chemical modifications to their impact on the enthalpy and entropy of binding. ITC directly measures the enthalpic and entropic contributions to binding, thus providing a unique window into the forces that determine the binding for specific ligands. New ITC instrumental developments, on the other hand, offer the unique opportunity of characterising a wide array of potential drug candidates, not only in terms of their binding affinity but also their binding enthalpy and entropy. These added dimensions can be expected to tremendously accelerate the drug development process.
References
- Lipinski, C.A. Physicochemical Properties and the Discovery of Orally Active Drugs: Technical and People Issues. in Molecular Informatics Confronting Complexity. Proceedings of the Beilstein Institute Workshop May 13th – May 16th, 2002 (ed. Kettner, M.G.H.a.C.) 59-78 (Logos Verlag, Berlin, Bozen, Italy, 2003).
- Ruben, A.J., Kiso, Y. & Freire, E. Overcoming roadblocks in lead optimization: a thermodynamic perspective. Chem Biol Drug Des 67, 2-4 (2006).
- Freire, E. A Thermodynamic Guide to Affinity Optimisation of Drug Candidates. in Proteomics and Protein-Protein Interactions: Biology, Chemistry, Bioinformatics and Drug Design (ed. Waksman, G.) 291-307 (Springer NY, 2005).
- Sarver, R.W. et al. Binding thermodynamics of substituted diaminopyrimidine renin inhibitors. Anal Biochem 360, 30-40 (2007).
- Schon, A. et al. Thermodynamics of binding of a low-molecular-weight CD4 mimetic to HIV-1 gp120. Biochemistry 45, 10973-80 (2006).
- Lafont, V. et al. Compensating enthalpic and entropic changes hinder binding affinity optimisation. Chem Biol Drug Des 69, 413-22 (2007).
- Cabani, S., Gianni, P., Mollica, V. & Lepori, L. Group contributions to the thermodynamic properties of non-ionic organic solutes in dilute aqueous solution. J. Solution Chem. 10, 563-595 (1981).
- Velazquez-Campoy, A., Kiso, Y. & Freire, E. The Binding Energetics of First and Second Generation HIV-1 Protease Inhibitors: Implications for Drug Design. Arch. Biochim. Biophys. 390, 169-175 (2001).
- Ohtaka, H. & Freire, E. Adaptive Inhibitors of the HIV-1 protease. Progr. Biophys. Mol. Biol. 88, 193-208 (2005).
- Ohtaka, H. et al. Thermodynamic Rules for the Design of High Affinity HIV-1 Protease Inhibitors with Adaptability to Mutations and High Selectivity Towards Unwanted Targets. Int. J. Biochem. Cell Biol 36, 1787-1799 (2004).
- Carbonell, T. & Freire, E. Binding thermodynamics of statins to HMG-CoA reductase. Biochemistry 44, 11741-11748 (2005).
- Wiseman, T., Williston, S., Brandts, J.F. & Lin, L.N. Rapid measurement of binding constants and heats of binding using a new titration calorimeter. Anal. Biochem. 179, 131-135 (1989).
- Straume, M. & Freire, E. Two-Dimensional Differential Scanning Calorimetry: Simultaneous Resolution of Intrinsic Protein Structural Energetics and Ligan Binding Interactions by Global Linkage Analysis. Anal. Biochem. 203, 259-268 (1992).
- Brandts, J.M., Brown, R.K., O’Brien, R. & Peters, W.P. ITC-Derived Binding Constants: Using Microgram Quantities of Protein. in Label-Free Biosensors: Techniques and Applications (ed. Cooper, M.) (Cambridge University Press 2007).
Dr Ernesto Freire
Faculty Professor, Johns Hopkins University, Baltimore
Ernesto Freire is the Henry Walters Professor of Biology at The Johns Hopkins University. He has been a member of the Department of Biology and Biophysics since 1986. He also holds a joint appointment in the Department of Biophysics and Biophysical Chemistry at the Johns Hopkins Medical School, and is a member of the Steering Committee of the Johns Hopkins Malaria Research Institute. Dr. Freire conducts research on HIV infection, malaria, genomic diversity, molecular recognition, and drug design. He has pioneered the development of new molecular design algorithms aimed at engineering drugs that exhibit extremely high affinity, selectivity against unwanted targets, and that maintain their effectiveness in the face of drug resistance mutations and genomic diversity. He is also developing novel thermodynamic-based algorithms aimed at engineering low molecular weight replacements for protein biologics. The author or co-author of over 200 publications and several patents, Dr. Freire is on the editorial board of Proteins: Structure, Function and Bioinformatics, Current Protein and Peptide Science and Chemical Biology and Drug Design. He was the recipient of the Stig Sunner Memorial Award for Outstanding Contributions to Thermochemistry and Thermodynamics in 1988, he was awarded a Johnson & Johnson Focused Giving Program Award in 1993 and was the recipient of the Microcal Prize for Outstanding Contributions to Experimental Thermodynamics in 2004. He has served on many scientific advisory committees for the National Institutes of Health and the National Science Foundation, and he holds memberships in the American Chemical Society, the Protein Society, and the Biophysical Society. Dr. Freire is an Honor Member of the Spanish Society of Biochemistry and Molecular Biology, and a member of the Academy of Sciences of Latin America.




