Applying ISO/TR 14644-21 – Airborne particle sampling techniques
23 January 2024 | By Particle Measuring Systems
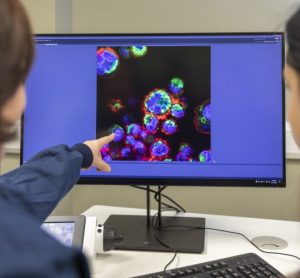
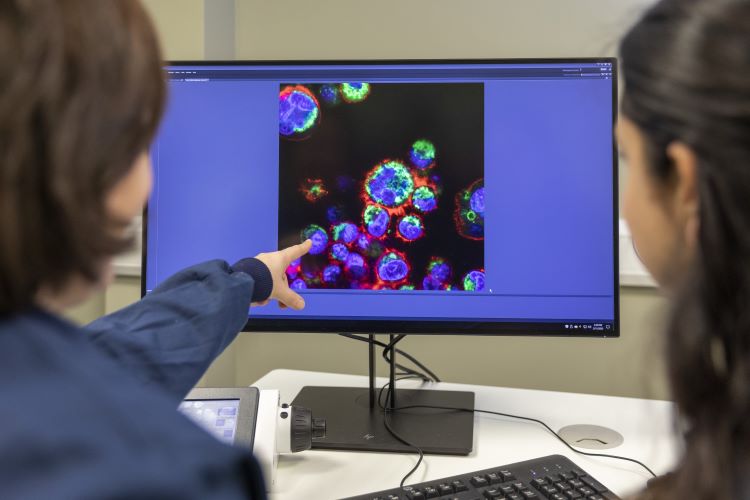
Watch this webinar as GMP expert Mark Hallworth discusses the difference between cleanroom classification and monitoring of the new ISO 14644 Technical Report 21 focuses in section 4.0 and 5.2.























![The GlaxoSmithKline headquarters building in Brentford, west London, June 2018 [Credit: Willy Barton / Shutterstock.com].](https://www.europeanpharmaceuticalreview.com/wp-content/uploads/GSK-3-300x278.jpg)
![The GlaxoSmithKline headquarters building in Brentford, west London, June 2018 [Credit: Willy Barton / Shutterstock.com].](https://www.europeanpharmaceuticalreview.com/wp-content/uploads/GSK-3-e1624534468322.jpg)
