The road ahead and how to get there
Posted: 22 August 2005 | | No comments yet
Ion channels are membrane spanning proteins with narrow hydrophilic pores that support the passive flux of inorganic ions across the cell membrane.
Ion channels are membrane spanning proteins with narrow hydrophilic pores that support the passive flux of inorganic ions across the cell membrane.
Ion channels are membrane spanning proteins with narrow hydrophilic pores that support the passive flux of inorganic ions across the cell membrane.
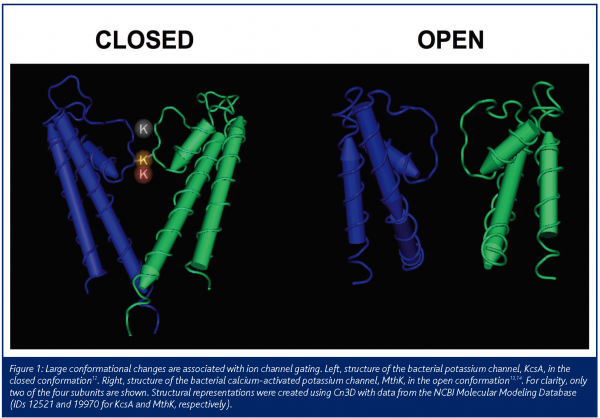
Channels are dynamic proteins which can exist in multiple states such as the open, closed and inactivated states and structural studies1 have shown substantial conformational differences between channel states (Figure1). Therapeutically used drugs often selectively bind to a specific channel state. Functionally, ion channels can be classified according to how they transition, or ’gate‘, between these states. Voltage-gated ion channels gate in response to changes in membrane potential whereas ligand-gated channels gate in response to the binding or unbinding of a ligand. In the open state, channels achieve remarkable transport efficiency as more than 1 million ions can pass through a single channel per second. Channels can also display exclusive ion selectivity, permitting some inorganic ions to pass but not others. To date, upwards of 300 different human ion channel genes have been identified yet merely 5 per cent of drugs marketed today target ion channels. During the past 15 years, major advances in both our understanding of the role of ion channels in disease, as well as ion channel screening technologies, have made this a molecular target class ripe for drug development.
A prerequisite in modern drug discovery is high throughput screening to facilitate the rapid evaluation of novel chemical entities. Until recently, high throughput assays for ion channels have relied on indirect methods of monitoring ion channel activity and thus compromised data quality for throughput. Examples of this include binding, ion flux and fluorescence based assays. These assays do not have the fidelity to distinguish detailed channel kinetics, a feature which is required to identify and characterise compounds with complex mechanisms of action such as gating modifiers and/or voltage, state, rate or use-dependent inhibitors. Such mechanisms of action are desirable for some ion channel drug targets since they can impart functional selectivity in vivo. For example, the Na+ channel antagonists lamotrigine and lidocaine preferentially block the channel in its inactivated state and the L-type Ca2+ channel antagonists nimodipine and nifedipine preferentially block the channel in its open state. The only technique with adequate sensitivity and the time resolution required for precise and direct measurement of ion channel activity and pharmacology is patch clamp electrophysiology. The recent introduction of high throughput electrophysiology instrumentation has revolutionised ion channel drug discovery. For the first time, we have the capacity to screen 100’s to 1000’s of compounds with the reliability and resolution of the patch clamp technique.
Historical view
Up until the past few decades, whole animal or tissue-based assays were used to discover ion channel modulators. If the physiology and pharmacology of the model tissue or organism matches the human condition, then this approach has the advantage that modulators are tested under conditions where the cellular activity and ionic concentrations are appropriate. The clear advantage of this approach is that compound optimisation is driven by data that may predict human clinical pharmacology of compounds. The substantial disadvantages of this approach are that throughput is unacceptably low and that, in some cases, a predictive animal model is not available. This can occur when the set of ion channel subtypes expressed in a human cell type of interest is not replicated in other species, or when studying some human pathologies, such as schizophrenia and depression, that are challenging to define in an animal model. This effective, but labour-intensive approach has produced ion channel modulators that are therapeutically effective due to state-dependent binding (e.g., calcium channel blockers used as antihypertensives) or tissue selectivity (sulfonylurea antidiabetics that block ATP-sensitive K channels in pancreatic beta-cells).
Application of in vitro biochemical assays marks the beginnings of a molecular approach to ion channel drug discovery starting more than 20 years ago. Assays measuring binding of radiolabeled ligands or isotopic ion flux enabled a target-based approach with a substantial improvement in throughput and a more efficient path for compound optimisation. Also, for the first time, random screening of compounds was feasible and novel lead classes could be readily identified. These approaches could be applied to cells or membranes derived from native tissues expressing the appropriate target channel. Limitations of these approaches include an inability, in some cases, to obtain adequate quantities of the target tissue and in most cases, use of non-human tissue sources, where channel expression may differ from human. Ligand binding experiments remain the most robust and reproducible assays for ion channel modulators and are still in widespread use, particularly in assays for undesired off-target activities including block of cardiac hERG potassium channels and cardiac sodium channels. Two significant limitations of ligand binding assays in target-oriented drug discovery are apparent. First, a specific ligand with the appropriate physical and pharmacological properties is a prerequisite. This limitation is most severe for projects involving novel channel types that have been identified in genomic screens or genetic studies; often these channels lack known pharmacological probes. Second, binding assays provide little information on the functional effects of compounds on the targeted channel and thus require subsequent analysis in a functional assay that provides additional mechanistic information. As an example, an assay measuring charybdotoxin binding to maxi-K channels in vascular smooth muscle2 was used to identify novel, potent and specific modulators of these channels with divergent functional properties, including subnanomolar blockers3,4 and a nanomolar activator5. Finally, ligand binding assays may fail to identify functionally active compounds or may identify compounds with no detectable functional activity. Despite these limitations, ligand binding assays remain an important component of some ion channel drug discovery programs and can be particularly useful in drug mapping studies with recombinant channels.
Ion flux assays have been used in drug discovery efforts for sodium, calcium, potassium and chloride channels. These assays monitor changes in ion concentration following modulation of channel activity; these changes are often small for physiologic ions, but the resolution can be substantially improved using permeant, radioactive isotopes. Such assays can be configured for medium-to-high throughput formats and provide linear measures of channel activity. Depending on channel type, triggering channel opening can be accomplished by adding a neurotransmitter, potassium to change the membrane potential, or a test compound. The poor temporal resolution of these assays hampers their application for channels that desensitise or inactivate. The introduction of recombinant ion channel expression techniques to drug discovery has reduced some of the problems associated with background fluxes encountered when using vesicles or cells derived from native tissues. Improvements in automation (Aurora Biomed) and detection of non-radioactive ions6 can eliminate the burdens associated with use of radioactive ions. Ion flux assays have neither the throughput of fluorescence plate-based assays nor the resolution and control of electrophysiological assays, so will find uses in situations where these alternatives cannot be applied.
Fluorescence assays run in high density plate formats have revolutionised ion channel drug discovery during the past ten years. The recent development of improved voltage-sensitive dyes7 along with indicator dyes for calcium and other ions allow the pharmacology of a wide variety of ion channels to be addressed in a high density format. In some cases, the cell lines and assays can be configured to detect state- or voltage-dependent channel modulation by controlling the membrane potential during the compound incubation period or by use of novel stimulation protocols, such as electrical field stimulation8. Some limitations exist. Membrane potential is a nonlinear, indirect measure of ion channel activity, so the activity of compounds in assays based on this parameter may shift from their intrinsic effects. Also, some compounds non-specifically interfere with cell activity or the dye systems. Fluorescence assays are an essential component of many current ion channel drug discovery programs, but do not negate the need for electrophysiological assays for confirmation and direct interrogation of functional effects of compounds. The development of automated patch clamp is beginning to make an important impact on how compounds are evaluated for drug discovery.
Automated patch clamp: current status
Several commercial automated patch clamp systems are now available that allow simultaneous recording of ion channel currents from multiple cells at once. As reviewed in detail in a previous article9, the systems fall into two broad categories. In one format, a chip with multiple wells is used for the asynchronous recording of many cells (presently up to 16) at once. These systems include the NPC-16p by Nanion, the QPatch-16 by Sophion and the PatchXpress 7000A by Molecular Devices Corp. Specialised chip fabrication permits high resistance (gigaohm) seals of the cell membrane with the substrate to achieve high quality data that rivals conventional patch clamp. Compared to conventional electrophysiology, the increase in throughput is typically 3-10 fold. Importantly, these devices allow continuous recording of channel activity. This feature allows assessment of the kinetics of drug interaction with the channel, an important parameter when measuring compound potency. These devices utilise integrated fluidics for the precise timing of drug addition, making them suitable for the study of ligand-gated channels. In a unique variant to conventional patch clamp, the Flyscreen 8500 by Flyion adds cells to the interior of conventional glass microelectrodes and uses standard patch clamp amplifiers and software to boost throughput approximately three fold.
The IonWorks HT & Quattro from Molecular Devices Corp. define the second category. Based on a 384-well microtitre plate, these systems yield up to a 100 fold enhancement in throughput compared to conventional patch clamp9-11. However, this increase in throughput is accompanied by a decrease in data quality. The substrate yields lower seal resistances and the electronics are streamlined, making these devices best suited for voltage-gated ion channels that express well in heterologous systems. Assays of ligand-gated ion channels are not possible and measurement of fast (rise time <1 ms) voltage-gated ion channels such as sodium channels are problematic with this platform. Nonetheless, this device provides the unprecedented ability to directly measure the activity of larger sets of compounds on an ion channel of interest. This throughput makes the IonWorks platform well-suited for lead optimisation, primary screening of directed compound libraries, secondary screening of hits from high throughput screening assays, and early safety pharmacology profiling. Currently, the lower throughput, gigaohm seal instruments and the higher throughput 384-well devices each have important roles in electrophysiological characterisation of novel compounds.
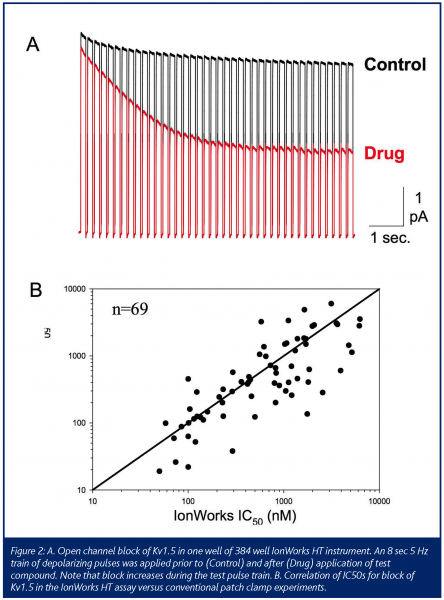
Given that state-dependence is an important property of the drug-channel interaction that influences therapeutic activity and safety, any automated electrophysiology device needs to be able to detect this. For voltage-gated channels, this requirement translates into flexible operator control of voltage as occurs in conventional patch clamp experiments. The automated devices have met this requirement to varying degrees. State-dependent channel block is clearly detectable with optimised voltage protocols in some assays (see Fig. 2A). In general, the 16-well devices provide higher flexibility than the 384 well platforms. In some cases, limitations in protocol design and voltage control may dictate whether a particular assay can be performed on a given device.
Automated patch clamp: needs and prospects
The application of these automated patch technologies has begun to change ion channel drug discovery. As our experiences with these instruments grow, several issues have become apparent that will need to be addressed in the next generation devices.
One issue that has come to the forefront is the correlation of compound potency between automated and conventional patch clamp. As shown in Figure 2B, the correlation can be quite good. However, when many compound structural classes are studied, it becomes clear that major discrepancies can exist. When discrepancies occur, the potency on the automated device is generally lower than by conventional patch. Several underlying factors can contribute in these cases. Since most devices employ a static well whereas conventional patch uses a continuously flowing bath, compound adsorption to the substrate and wall of the well can contribute. Until the underlying causes are more completely understood and remedies are in place, parallel assays will always be needed.
Another area ripe for further development is software. Some systems, like the plate-based devices, would benefit from more flexible control of stimulus voltage. Most importantly, the ability to efficiently analyse, create reports and store the data has not kept pace with the increase in throughput. Now many users rely on custom-designed or third-party software to fill this need. Once the technological milestones have been reached, these issues will certainly come to the forefront.
In the case of the mircotitre-based systems, improvements in data quality would definitely be welcomed. Substrates that increase seal resistance even two-fold would improve data quality significantly. Fast voltage control and data acquisition coupled with continuous recording would make the mircotitre-based format extremely powerful.
Given time, these needs will certainly be addressed. The hope of studying the state-dependence of numerous compounds by automated patch clamp is rapidly becoming a reality. The bright future of ion channel drug discovery and safety screening owes much to the promise of these emerging technologies.


References
- MacKinnon R (2004). Nobel Lecture. Potassium channels and the atomic basis of selective ion conduction. Biosci Rep. 24, 75-100.
- Vazquez J, Feigenbaum P, Katz G, King VF, Reuben JP, Roy-Contancin L, Slaughter RS, Kaczorowski GJ & Garcia ML (1989). Characterization of high affinity binding sites for charybdotoxin in sarcolemmal membranes from bovine aortic smooth muscle. Evidence for a direct association with the high conductance calcium-activated potassium channel. J Biol Chem. 264, 20902-20909.
- Knaus HG, McManus OB, Lee SH, Schmalhofer WA, Garcia-Calvo M, Helms LM, Sanchez M, Giangiacomo K, Reuben JP, Smith AB, 3rd, Kaczorowski GJ & Garcia ML (1994). Tremorgenic indole alkaloids potently inhibit smooth muscle high-conductance calcium-activated potassium channels. Biochemistry. 33, 5819-5828.
- Garcia ML, Kaczorowski GJ & McManus OB (2003). US Patent 6,545,036.
- McManus OB, Harris GH, Giangiacomo KM, Feigenbaum P, Reuben JP, Addy ME, Burka JF, Kaczorowski GJ & Garcia ML (1993). An activator of calcium-dependent potassium channels isolated from a medicinal herb. Biochemistry. 32, 6128-6133.
- Terstappen GC (1999). Functional analysis of native and recombinant ion channels using a high-capacity nonradioactive rubidium efflux assay. Anal Biochem. 272, 149-155.
- Gonzalez JE & Tsien RY (1997). Improved indicators of cell membrane potential that use fluorescence resonance energy transfer. Chem Biol. 4, 269-277.
- Maher MP & Gonzalez JE (2004). US Patent 6,686,193.
- Trezise DJ (2005). Patch clamp electrophysiology steps a gear. European Pharmaceutical Review. 2, 69-73.
- Schroeder K, Neagle B, Trezise DJ & Worley J (2003). Ionworks HT: a new high-throughput electrophysiology measurement platform. J Biomol Screen. 8, 50-64.
- Kiss L, Bennett PB, Uebele VN, Koblan KS, Kane SA, Neagle B & Schroeder K (2003). High throughput ion-channel pharmacology: planar-array-based voltage clamp. Assay Drug Dev Technol. 1, 127-135.
- Doyle DA, Morais Cabral J, Pfuetzner RA, Kuo A, Gulbis JM, Cohen SL, Chait BT & MacKinnon R (1998). The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science. 280, 69-77.
- Jiang Y, Lee A, Chen J, Cadene M, Chait BT & MacKinnon R (2002). Crystal structure and mechanism of a calcium-gated potassium channel. Nature. 417, 515-522.
- Jiang Y, Lee A, Chen J, Cadene M, Chait BT & MacKinnon R (2002). The open pore conformation of potassium channels. Nature. 417, 523-526.




