Serialisation reconciliation: a blind spot in your finished product’s manufacturing compliance?
Posted: 30 March 2017 | | 1 comment
Pharma serialisation expert, Pasi Kemppainen, gives us his insights on how serialisation reconciliation can ensure product integrity and patient safety…

The purpose of the pharmaceutical reconciliation process is to ensure that all materials used for finished products have been correctly accounted for and no errors have occurred. The reconciliation helps to avoid releasing a non-conforming product, and is the very foundation for ensuring product integrity in the product supply chain and, ultimately, patient safety.
However, pharmaceutical finished goods manufacturing is already changing extensively due to global serialisation and traceability regulations. These regulations require that each and every medicine sales package requires unique numbering and traceability throughout the supply chain – right up to the patients. Obviously, implementing these new capabilities in the finished pharma manufacturing should help improve the medicinal product’s integrity and comply with the current reconciliation requirements specified in the regulatory GxP (Good Practices) guidelines, especially by the US Food and Drug Administration (FDA) and European Medicines Agency (EMA).
Pharma serialisation reconciliation ensures that the serial numbers are used correctly for finished products with traceability in each phase of the pharmaceutical packaging process; beginning with the creation of serial numbers, and ending with supplying the finished serialised products to the market.
The current GMP (Good Manufacturing Practices) guidelines do not directly address the reconciliation of the serial numbers in the packaging process. However, they do require that all drug product production and control records, including those for packaging and labelling, shall be reviewed and approved by the quality control unit in order to determine compliance with all established, approved written procedures before a batch is released or distributed. In particular, the GMP guidelines require controlling the quantities and reference number or identification of all printed packaging materials, as well as the quantities of obtained product, in order to provide adequate reconciliation.


This means that, from a GMP perspective, as the serial numbers are part of the control records and identification of printed materials, they should be managed and reconciled in a similar way to the current finished products material reconciliation.
In addition to GMP guidelines for pharmaceutical product production and packaging, regulators have also set out handling responsibilities and principles throughout the pharmaceutical supply chain, including Good Distribution Practices (GDP) and Good Storage Practices (GSP). These include, but are not limited to, storage, distribution, transportation, packaging, labelling, documentation and record-keeping practices. For example, in a case of recall GDP states that the progress of a recall process should be recorded and a final report issued, which includes the reconciliation of delivered and recovered quantities of products. In a case of serialised products, in the future there will certainly be a requirement for the serial number reconciliation in the final report.


GDP and GSP guidelines extend the management and reconciliation of serial numbers beyond manufacturers’ own control and will therefore require timely and correct reporting about the changes in the product status between Marketing Authorisation Holders (MAH) and Contract Manufacturer Organisations (CMO), and e-pedigree services between supply chain partners. Therefore, when planning the serialisation implementation and quality processes, it is important that the serialisation reconciliation processes are planned also to support the regulatory and business reporting requirements beyond the current GMP guidelines.
Pharma serialisation reconciliation process
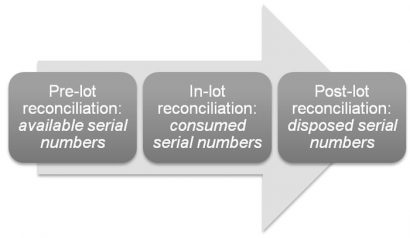
The serialisation reconciliation process can be divided into three phases based on the batch packaging execution process (lot):
- Pre-lot reconciliation for the reservation, creation and provision of serial numbers to the packaging lines. The pre-lot phase defines the total amount of available serial numbers used for the packaging
- In-lot reconciliation covers the serial number usage for finished products and shipping cases/pallets (aggregation), as well as in-line disposal and quality management sampling. The in-lot phase defines the actual serial number consumption in the packaging. In addition, EMVS (European Medicines Verification Service) reporting needs to be done as part of the in-lot reconciliation
- Post-lot reconciliation covers the changes with the serialised products and aggregation after packaging completion but prior to the change of custody, e.g. warehouse damage control and quality control sampling.

Serialisation reconciliation process in the batch packaging execution
Pre-lot reconciliation: getting ready for the serialisation
In the pre-lot phase there’s a difference between whether the products are pre-printed or printed in-line. As the actual allocated serial numbers in the packaging usually represent less than received serial numbers, it is important to manage the inventory of reserved (used) and unreserved (unused) serial numbers. For example, in the case of CMOs, if the serial numbers are being received from the Marketing Authorisation Holder (MAH), there can be a requirement to also report the unused serial numbers, rather than just those used in production.
If required, the yield for the pre-lot reconciled serial numbers can be calculated as follows:


The ‘allocated serial numbers’ is basically the total planned lot size, and the ‘total serial numbers’ is the number of serial numbers reserved for the lot. The information about the amount of allocated and total serial numbers is usually available in the manufacturer’s serialisation repository system.
In-lot reconciliation: ensuring compliance
The in-lot reconciliation is obviously the most important phase from the quality management and GMP compliance point of view. The in-lot process allocates the received serial numbers for serialising the trade items and for aggregation, when required. In this process the serial numbers can be disposed for several reasons due to the technical problems, material problems and quality management.
The reconciliation can be calculated based on the number of serial numbers that were successfully activated (commissioned) with all disposed serial numbers. This total amount of utilised serial numbers will then be compared to the allocated pre-lot serial numbers. As mentioned, MAHs may require reporting all serial numbers including the unused allocated pre-lot serial numbers.
If required, the yield for the in-lot reconciled serial numbers can be calculated as follows:
![]()
![]()
The in-lot serial number changes are recorded either in the serialisation vendor plant, or site server, or in the manufacturer’s centralised serialisation repository.
For the products mandated by the EU Falsified Medicines Directive, there is a completely new and important process that needs to take place before the lot can be released: reporting to the European Medicines Verification Service. This needs to be done after the in-lot reconciliation but before the lot release by the Qualified Person. The reason for this is that the medicinal products cannot be released to the market before they are successfully reported to EMVS, and therefore the successful EMVS reporting needs to be included in the lot release report in addition to the correct serial number reconciliation.
Post-lot reconciliation: supplying products to the market
The post-lot reconciliation covers the changes that occur after the packaging stage, but while the serialised products are still in the manufacturer’s custody – e.g. in staging, transit and warehouse. In an ideal case, there would be no need for the post-lot reconciliation with corrective actions such as removing the damaged packages and taking QA samples. However, any such changes need to be recorded and potentially also reported to MAHs and regulators.
If required, the yield for the post-lot reconciliation can be calculated as follows:
![]()
![]()
It is important to notice that after the post-lot reconciliation and reporting in the manufacturer’s repository and EMVS, the serial numbers are not changed or deleted even in the case of eradication and disposal. For example, in a case of replacing a trade item or even a logistical unit (disaggregation) in the lot, a disposed serial number is decommissioned and a new replacing serial number created, commissioned and aggregated. Only the dispositions (states) of the serial numbers can change and will be reported to the manufacturer’s serialisation repository and EMVS.
When the finished products are being shipped from the manufacturer to the supply network, the maintaining responsibility of the serial numbers states in EMVS will change and the states can be changed by third-party stakeholders, e.g. parallel distributors, wholesalers, national competent authorities and point of dispense network (mainly pharmacies and hospitals).
In any case, MAHs need to still be able to maintain the in-lot serialisation data (serial numbers, new dispositions and related timestamps) for one year after expiry of the lot, or at least five years after certification of the lot by the Qualified Person, whichever is the longer.
Turning challenges into possibilities
Serialisation reconciliation can bring a wide variety of challenges if not properly specified and planned before implementing the serialisation in production. This is particularly the case regarding alignment with the existing packaging QA and QP lot/batch release procedures during and after the production, which is essential.
For the highest level reconciliation accuracy and supply network manageability, it is highly recommended to use aggregation. This ensures more cost-efficient supply network operations and traceability on a case and pallet level. For example, finding a lost or extra serial numbers (more packed trade items than used in-lot serial numbers) would require extensive rework before the lot can be released. Also, as the EU FMD mandates the risk-based verification in the supply network, many wholesalers will most likely require aggregation to ensure efficient verification based on the case or pallet level serial numbering.
The following are some recommendations that can help with the complex process of managing the serialisation reconciliation:
Pre-lot reconciliation | Specify and document the pre-lot serialisation process reconciliation requirements in the serialisation system and repository URSs |
Specify the serialisation pre-lot reconciliation standard operating procedures with cross- organisational responsibilities | |
Specify the pre-lot requirements and processes (e.g. acquiring the serial numbers and ranges) with MAHs and CMOs | |
In-lot reconciliation | Align the serialisation reconciliation process with the existing material reconciliation standard operating procedures for the GMP compliance |
For the highest level reconciliation accuracy and rework manageability, implement aggregation | |
Specify the in-lot serialisation reconciliation process, standard operating procedures and cross-organisational responsibilities | |
Ensure that the serialisation systems can provide the required information of the reconciliation process with the potential deviations (lost, orphan, etc.) for reporting | |
Include the EMVS reporting in the lot/batch release process and reporting | |
Include the in-lot reconciliation deviations in the Failure Mode and Effects Analysis (FMEA) mitigation plan | |
Post-lot reconciliation | Centralise the post-lot serialisation data management with a serialisation repository |
Specify the post-lot serialisation reconciliation process, standard operating procedures and cross-organisational responsibilities | |
Use the serialisation data to support operations and business analytics (e.g. OEE, quality metrics, warehousing rework, recall and chargeback management |





good for Good MP