Concurrent Evaluation of both Compendial and Rapid Methods (ATP Bioluminescence) for Monitoring Water Quality in Pharmaceutical Manufacturing
Posted: 29 May 2009 | Dr Eric Bagur, Bacteriological Quality Control Manager, Bristol-Myers Squibb | No comments yet
Much has been published elsewhere about the limitations of the compendial microbiological methods and the needs of current Pharmaceutical manufacturing, in particular related to the adoption of PAT as a quality tool1-4. Rapid Microbiology is seen by many as a tool for addressing a number of these limitations5,6. Rather less has been published in terms of case histories from users demonstrating how to apply rapid methods to solve specific problems arising from the use of traditional microbiology. This paper addresses this gap by presenting studies based on experience gained from utilising a rapid method based on ATP bioluminescence (the PallchekTM Rapid Microbiology System).
Much has been published elsewhere about the limitations of the compendial microbiological methods and the needs of current Pharmaceutical manufacturing, in particular related to the adoption of PAT as a quality tool1-4. Rapid Microbiology is seen by many as a tool for addressing a number of these limitations5,6. Rather less has been published in terms of case histories from users demonstrating how to apply rapid methods to solve specific problems arising from the use of traditional microbiology. This paper addresses this gap by presenting studies based on experience gained from utilising a rapid method based on ATP bioluminescence (the PallchekTM Rapid Microbiology System).
Much has been published elsewhere about the limitations of the compendial microbiological methods and the needs of current Pharmaceutical manufacturing, in particular related to the adoption of PAT as a quality tool1-4. Rapid Microbiology is seen by many as a tool for addressing a number of these limitations5,6. Rather less has been published in terms of case histories from users demonstrating how to apply rapid methods to solve specific problems arising from the use of traditional microbiology. This paper addresses this gap by presenting studies based on experience gained from utilising a rapid method based on ATP bioluminescence (the PallchekTM Rapid Microbiology System).
Water Analysis – Limitations of the current method
Historical practice for monitoring water systems has been based on the heterotrophic plate count. In this method:
- Water samples are filtered through 0.45mm membranes
- The membranes are plated in duplicate onto R2A agar and incubated (5 days, 30°C)
- Colonies are counted after 5 days and the count is then compared with the alert and action limits established for the water system being monitored (see Figure 1).
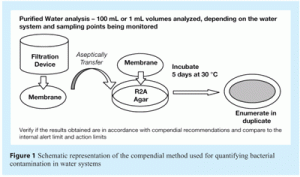

The compendial method has the advantages of being relatively easy to perform because of the low materials required and a moderate to low cost. The major disadvantages of this method are the time delay between performing the test and obtaining the results and the possibility that stressed organisms may not be detected.
The delay between obtaining an OOS result and taking corrective action is a major problem. It may cause underestimation of the real contamination level, firstly because additional growth may occur in the water system during the incubation period (particularly of stressed organisms, which may be slow growers). Underestimation of contamination level may occur resulting in inappropriate, and thus ineffective, corrective action.
Water systems studied
Two distribution water systems at the Guyenne plant (existing plant) and two at the Gascogne plant (new plant) were both monitored in this study.
In the old plant, a total of 27 sample points are routinely analysed, including eight sample points from production loop and 19 sample points from the distribution loop.
In the new plant, a total of 72 sample points are routinely analysed, including 42 sample points from production loop and 30 sample points from distribution loop.
Table 1 shows action levels, which are based on the compendial methods. These are based on the historical data.


The water quality monitoring action level established for the two plants are different. They were fixed at different levels, according to comparability, between produced and distributed purified water quality and historical monitoring data.
ATP Bioluminescence as a rapid method
Adenosine triphosphate (ATP) bioluminescence is a widely-used technology, e.g. in the Food industry, as a quality monitoring tool7,8. It is a well accepted technology and already accepted as a sensitive method for water testing9 having gained regulatory acceptance for the release of pharmaceutical products as well as for monitoring pharmaceutical water9.
Quantitative ATP bioluminescence measurements can be made within minutes. However, these measurements are limited in terms of detection sensitivity. This is because they directly measure the ATP content of any microorganisms in a sample. This limits sensitivity to around 100-1000 cfu.
Qualitative and semi-quantitative measurements can be used to demonstrate the presence or absence of very low levels of contaminating microorganisms within 24 hours (qualitative measurements) or to estimate the level of contamination (semi-quantitative measurements). Further details of these methods are provided in the Methods and Materials section.
Materials and Methods
Test system description
The technology evaluated was the PallchekTM Rapid Microbiology System (see Figure 2).
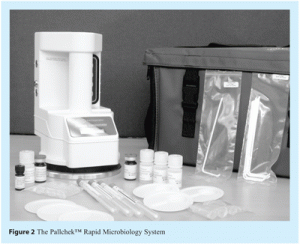

The system consists of a handheld luminometer that detects microbial contamination based on measurement of ATP. The luminometer seals onto an aluminum test plate, to create a light-tight measuring system using a flexible gasket that surrounds the measuring chamber. During a measurement, this gasket is sealed against the aluminum plate using vacuum, provided by a vacuum pump within the luminometer.
When performing a test, samples are placed onto disposable plastic trays which fit onto the aluminum test plate. Reagents are added to the sample to enable ATP measurement.
The first reagent is an extractant, which lyses any microbial cells present in the sample to release intracellular ATP. The second reagent, added after the lysis step, is a luciferin-luciferase substrate/enzyme reagent, incorporating material extracted from the firefly, Photinus pyralis. This enzyme/substrate reaction generates photons in proportion to the level of ATP present in the sample analysed. Photons emitted by the sample are measured using the PallchekTM luminometer. This instrument contains a photo-multiplier and associated circuitry for photon detection and amplification. Results are displayed on a liquid crystal display on this luminometer as Relative Light Units (RLU). One RLU represents a count of one photon per second.
Microbial cells contain relatively constant levels of ATP. This level depends on the type of microorganism, cell size, nutrient conditions, phase of the growth cycle and stress. Therefore any direct relationship between microorganism count and RLU reading must be demonstrated using standardised samples. This point is covered in more detail in the Results and Discussion section.
Normally, the system is used in conjunction with sample collected using membrane filtration. For a sample concentrated on membrane, measurements can be made in two main ways:
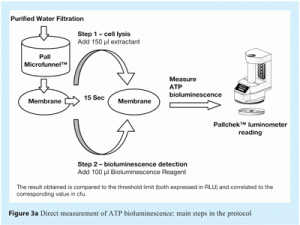
- Direct measurement of ATP in which a sample is collected and immediately analysed using the PallchekTM Rapid Microbiology System. This method has a detection limit of around 100-1000 cfu, based on the amount of ATP typically contained in bacterial cells and the limit of detection achievable with the reagents and luminometer used. This method provides a result about one minute after sample filtration. Figure 3a shows the main steps in this method.

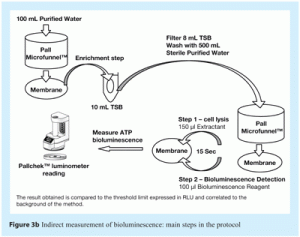

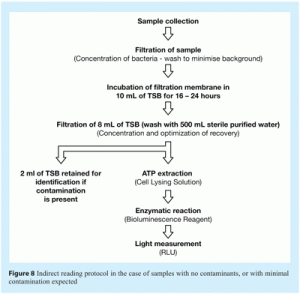
- Indirect measurement of ATP in which the filtered sample is inoculated into 10mL of liquid culture media and incubated overnight (enrichment step). Following incubation, 8mL of the liquid culture media is analysed. This method can detect down to one cfu and provides results within 24 hours. Figure 3b, shows main steps in this method.
Using samples collected by membrane filtration has several advantages. These are:
- The membrane can be washed with appropriate solutions to remove any sample components that might interfere with detection.
- Any microorganisms present in the sample are concentrated on the membrane surface.
- This maximises the amount of light signal proximal to the photomultiplier tube in the luminometer.
- Contact between the reagents used for ATP measurement and any microorganisms present in the sample is maximised.
Preliminary Study
As with any new technology, demonstration of system capability and Performance Qualification are critical components of the successful introduction of Rapid Microbiology technology. This section describes how this was undertaken for the PallchekTM Rapid Microbiology System. The steps covered in this section are:
- System suitability tests, including background measurements and confirmation of reagent performance
- Correlation of RLU with ATP
- Correlation of RLU with cfu
- Threshold value determination
(i) System suitability tests
To ensure that the system is functioning correctly, System Suitability Tests were performed at the beginning of every measurement session, following the supplier’s recommendations. These tests were as follows:
- Background measurements – Demonstration of consistent, low, background readings minimises the possibility of recording false positive results. Background is measured in two stages, first for the instrument and second for the reagent.
To determine instrument background, the luminometer is placed on the aluminum test plate supplied with the instrument and a background reading taken. This should be < 20 RLU.
To determine the reagent background, a plastic sample tray is placed on the aluminum test plate. 150 µL of extractant is added to this tray, followed by 100 µL of reconstituted bioluminescence reagent. A measurement is then taken within five seconds. The reading obtained should be < 80 RLU.
- Confirmation of reagent performance – This test acts as a positive control demonstrating that a specific concentration of ATP gives a reading within defined, expected, limits.
A plastic holder is placed on the aluminium test plate. 100 µL of a 10-9 M solution of ATP standard (produced using the manufacturer’s ATP Correlation Kit) is added to the sample holder, followed by 100 µL of reconstituted bioluminescence reagent and the reading is taken.
The reading obtained is typically between 105 RLU and 106 RLU when the Pall High Sensitivity Reagent Test Kit is used.
(ii) Correlation of RLU with ATP
The production of an ATP Correlation Curve is a basic procedure used for operator training and comparison of user measurements with those obtained by the manufacturer during system validation. The procedure also allows the user to identify any drift in system performance and to qualify and compare the performance of new batches of reagents with those previously utilised.
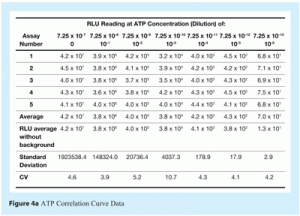
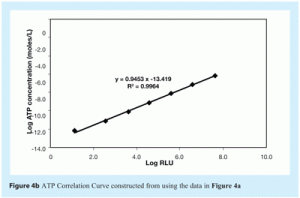
In this method, six serial, ten-fold, dilutions of the ATP standard solution supplied by the manufacturer are made. For each of these dilutions, the RLU values are measured five times. The data obtained are used to construct a graph by plotting logarithmic values of ATP concentrations on the x-axis and logarithmic values of RLU readings on the y-axis. An example is presented in Figures 4a and 4b (see Results and Discussion).


(iii) Correlation of RLU with cfu
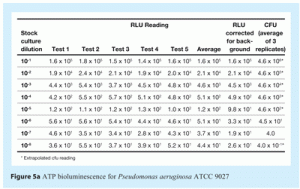
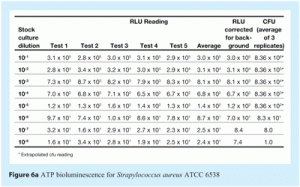
The second main step in system qualification is correlation of RLU readings and cfu measurements. The two main objectives of this study are characterisation of sample background and establishment of the relationship between RLU readings and cfu measurements for specific relevant microorganisms. The method used is as follows:
- Control samples are inoculated with microorganisms
- These samples are serially diluted in ten-fold steps, to produce samples covering a range of microorganism concentrations from roughly 101 to 108 cfu
- The RLU reading of each serial dilution is measured five times
- In parallel, samples are plated onto agar and incubated. These samples are used to determine the number of cfu for each sample analysed
- A correlation curve is created by plotting the logarithmic values of the cfu count (from the agar plates) on the x-axis and the logarithmic values from the RLU measurements on the y-axis.
In addition, five control samples (no microorganisms present) were tested, to determine background.
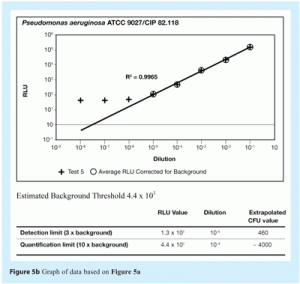
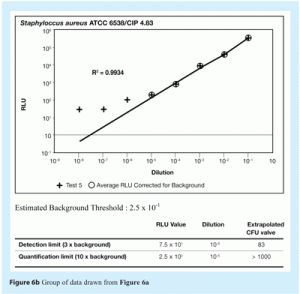
Representative correlation curves are shown in Figures 5a, 5b, 6a and 6b.




(iv) Threshold determination
The detection limit for ATP bioluminescence was set at three times the background level. The detection limit represents the RLU value above which RLU readings can be considered different from background.
The quantification limit for ATP bioluminescence was set at 10 times the background level. The quantification limit represents the RLU value above which RLU readings give an accurate estimation of the number of microorganisms present in a sample.
The RLU values established for the detection and quantification limits were converted to the corresponding cfu values using the correlation curve between RLU and cfu just described. This conversion provided the number of microorganisms that corresponds to these two RLU values.
In accordance with the International Committee for Harmonisation’s recommendations, a threshold limit was fixed at five times the background for all data generated using the different microorganisms tested. This corresponds to at least 1000cfu.
Water system monitoring
This section describes the methods used for water system monitoring. It covers:
- Sample background measurement
- Semi-quantitative measurement procedures based on direct ATP measurement
- Qualitative measurement procedures, based on indirect ATP measurement.
As mentioned previously, the method used for monitoring depended on the quality of the water being monitored.
(i) Sample background measurement
Sample background is measured for uncontaminated control water samples that have been demonstrated to contain no microbial contamination using compendial methods. For this study, control samples used for background measurement were representative of the water production systems being monitored.
These samples provided measurements that define the threshold RLU value above which readings indicate the presence of microbial contamination. The method used to analyse these samples is shown in Figure 7. The threshold limit set based on the RLU readings obtained for these samples was five times the background RLU measurements obtained.
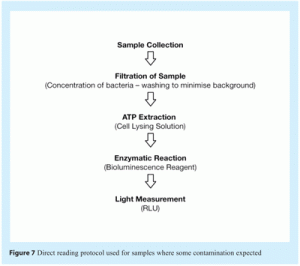

The RLU values obtained for these blank control samples should be < 150 RLU when filtration of the samples is performed using Pall® GN-6 Metricel® membranes. Other membranes may give different background values. In this case, the threshold value must be adjusted accordingly.
Several tests were performed, with different membrane washing volumes, to set up the lowest and reproducible RLU sample background value.
Blank controls are repeated during each routine test in order to reevaluate the threshold limit according to environmental conditions of testing.
(ii) Semi-quantitative procedure based on direct ATP measurement
This method was utilised when historical data demonstrated that some microbial contamination was likely to be present in water samples. In this procedure, water samples are collected and immediately analysed, providing a result within about one minute after sample filtration. This procedure is shown schematically in Figure 8.

The main steps in this method are:
- Water sample collection and filtration of three aliquots of this sample. The volume of these samples is based on the expected microbial contamination level (see Table 1).
- Immediate measurement of ATP bioluminescence values for these samples.
- Comparison of the RLU reading obtained against the threshold limit determined previously. This reading is reported as presence (+) or absence (-) of contamination, based on a RLU reading either above or below the threshold value previously determined.
Table 2 summarises the relationship between sample volume, presence (+) or absence (-) result obtained and the contamination range (number of cfu contained in the sample) that can be inferred, based on correlation of RLU to cfu. Note that this table is based on a study performed during methods development, using pure ATCC cultures, not with actual contaminated samples from the water systems studied. The objective of this table is to demonstrate how actual data can be ascribed probable contamination levels, based on RLU measurements.
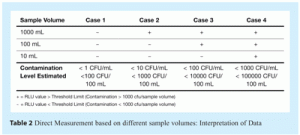

The advantage of direct measurement is that a real time result (data within 30 minutes) can be achieved and the method is fast and simple to perform. In addition, analysis of several sample volumes allows rapid estimation of contamination level and quickly identifies an OOS result, thus minimising the possibility of recording false positive or negative results.
The main disadvantage is sensitivity, limiting the method to water systems where there is typically a relatively high level of microbial contamination (> 1000 cfu/sample) present. For systems that deliver water quality at or near to few cells, typically ‘Water for Injection’ standards or highly ‘Purified Water’ systems, a different approach, described in the next section, is required.
(iii) Qualitative procedure based on indirect ATP measurement
Where the expected microbial contamination level in water samples is very low (0-100 cfu/100mL) semi-quantitative direct measurements are not suitable. This is because the RLU reading in this very low contamination range is at or below the quantification limit established during system qualification. A different approach must be used to determine if water quality is within acceptable limits.
The indirect method, described in Figure 3b, is a presence/absence method that measures ATP based on an enrichment step. This enrichment step involves membrane filtration of the sample and overnight incubation of the analysis membrane in liquid media. This step promotes microbial growth and thus amplification of the ATP bioluminescence signal obtained.
In this method, a water sample is collected and diluted twice in ten-fold serial dilutions, to provide 100mL volumes of undiluted samples and of samples at dilutions of 1/10 and 1/100. Each dilution is then filtered using a 47mm diameter analysis membrane. These membranes are aseptically transferred into 10mL volumes of liquid media. After overnight incubation, 8mL samples of this media are filtered through 47mm diameter analysis membranes. The ATP bioluminescence value of these membranes is measured, as described previously.
The background value of the method is established by analysing several control samples uninoculated with bacteria. Using the same approach as described for the semi-quantitative method, a threshold limit is set based on the mean background reading obtained. This threshold limit is set at five times the background.
A RLU result higher than the threshold limit represents a positive result (+) and means that the sample analysed contains at least 1 cfu. A RLU result lower than the threshold limit represents a negative result (-) and means that sample contains less than 1 cfu.
The data in Table 3 summarises the possible range of results achievable after 24 hours of incubation. This table demonstrates how the pattern of presence (+) or absence (-) results obtained for the three different dilutions can be used to estimate the contamination level in a sample, in the range of < 1 cfu/100mL to > 100 cfu/100 mL. Note that this table is based on a study performed during methods development, using pure ATCC cultures, not with actual contaminated samples from the water systems studied. The objective of this table is to demonstrate how actual data can be ascribed probable contamination levels, based on RLU measurements.


This method is very successful for a water system where very low microorganism contamination levels are expected.
The advantages of this method is that it provides a much more rapid alert compared to the compendial method, particularly when the compendial method is performed using R2A agar, where it is not possible to obtain results in less than five days.
Results and Discussion
Figure 4 shows a representative correlation curve of ATP concentration versus RLU readings. This demonstrates a very good correlation coefficient (r2) of 0.9964 between the two sets of data. As discussed in the Materials and Methods section, construction of ATP correlation curves represent a very good training tool for this type of ATP bioluminescence method. They also allow the user to assess any drift in instrument or reagent performance, to compare reproducibility of system performance between different operators, reagents and instruments and to assess method ruggedness and robustness by deliberately varying critical aspects of the method, such as sample and reagent volumes. Reproducibility, ruggedness and robustness are critical factors (identified with others) that are recommended to be part of new technology assessment in United States Pharmacopeia8 and European Pharmacopoeia9.
Figures 5 and 6 show representative data that demonstrate the correlation between RLU and cfu for two microorganisms, Staphylococcus aureus (ATCC 6538) and Pseudomonas aeruginosa (ATCC 9027). Note that these curves are for pure cultures. They may not be representative of data obtained using natural isolates from a manufacturing environment, since environmental stresses may well reduce the amount of ATP present in any recovered micro-organisms.
Representative data obtained from microbiological monitoring of the water systems in the BristolMyersSquibb plants at Gascogne and Guyenne are presented in Tables 4 and 5. Note that this part of the study was performed using the ATP bioluminescence method in parallel with the compendial method. Sampling methods and sampling times were different for both compendial method and bioluminescence method. In addition, tests were performed during validation of the loop (just after sanitisation or before the start of production) which makes it likely that any microorganisms present were subjected to additional stress.
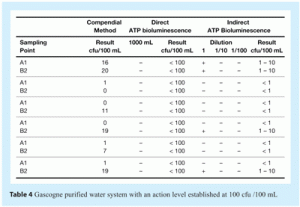

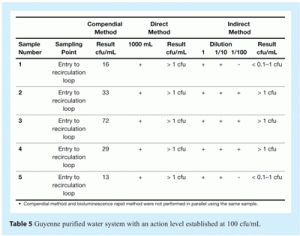

Despite the differences between the compendial and alternative methods, the results obtained with both were comparable (see Tables 4 and 5). These data show that the direct measurement method is better for the evaluation of contaminated samples with an action level of around 100cfu/mL. For the direct method, a sample volume of 100ml would be more appropriate to determine if the contamination level was above or below an alert level of 10cfu/ml, which is a more stringent alert level than the actual one (20cfu/mL).
Results obtained with the ATP bioluminescence method (using the indirect measurement of ATP) show a ten fold difference compared with the results obtained using the compendial method. The difference may be due to differences in sampling and testing conditions between the two methods but may also reflect the fact that ATP bioluminescence can detect stressed bacteria that may not be detected using the compendial method. In any case, these data demonstrate good overall correlation between the compendial method and the ATP bioluminescence method, namely:
- Gascogne plant: when the compendial method gives a result of > 10cfu, the bioluminescence method gives a result between 1-10cfu. From analysis of three different sample volumes, the ATP bioluminescence method can demonstrate whether the contamination level is above or below an alert level of 10cfu/100ml.
- Guyenne plant: the results from indirect measurement of ATP are not well-suited to contamination level of samples.
This study was an evaluation of the bioluminescence method, not a validation of the method. The results obtained helped to determine the appropriate analysis method, based on the contamination level of each water system. This study also shows that wild microorganisms do not behave in the same way as pure cultures and that the analysis method must be suited to the actual contamination level of any samples analysed.
Conclusions
The PallchekTM Rapid Microbiology System is a sensitive method for detecting microbial contamination. To obtain consistent and meaningful results, it is critical that:
- Accurate and aseptic techniques are used during the preparation, storage and use of reagents.
- Test procedures are standardised and followed exactly the same way every time that a test is performed.
- System suitability and other appropriate control experiments are performed.
- Successful operator training and continuing competency are confirmed using techniques such as the ATP Correlation Curve.
If these steps are followed, the method can be quickly implemented in routine Quality Control analysis.
This method allows simple interpretation of the results obtained and application of productive and fast tests. Application of the system as a water quality monitoring tool allows several important rapid actions to be implemented in a manner far more compliant with the objectives of PAT than is possible with the compendial methods.
These actions include:
- Resampling and analysis of purified water when an alert, action or specification limit is exceeded, allowing proactive corrective action to be taken.
- Quick negation or confirmation of an OOS result.
- Early alert to manufacturing to take the water system out of use as a safety measure, based on a valid OOS result (and to sanitise it as a precaution).
- Rapid analysis of subsequent water samples during system restart after a process excursion.
Acknowledgements
The authors wish to acknowledge the support of Catherine Piton, Lucia Ceresa and Peter Ball (Pall Life Sciences) during this study and the preparation of this paper.
References
- Miller, M. (2005). The impact of Process Analytical Technology, cGMP’s for the 21st Century and other regulatory and compendial initiatives on the implementation of Rapid Microbiological Methods. Encyclopedia of Rapid Microbiological Methods, Volume I, pages 195-295. Published by Davies Healthcare International Publishing LLC. ISBN 1-930114-85-0.
- Johnson, R. (2007). A “PAT” on the back for Rapid Microbiological Methods. European Pharmaceutical Review, Issue 4, pages 84 – 88
- Newby, P.J., G. Dalmaso, S. Lonardi, B. Riley, P. Cooney and K. Tyndall (2004). The introduction of qualitative rapid microbiological methods for drug product testing. Pharmaceutical Technology, Sept 1 issue, pages 6-12.
- Riley, B.S. (2006). Rapid Microbiology Methods in the Pharmaceutical Industry. American Pharmaceutical Review 7 (2): 28-31.
- Dostalek, P.and T. Branyik (2003). Prospects for Rapid Bioluminescent Detection Methods in the Food Industry – a Review. Czech Journal of Food Sciences 23 (3): 85-92.
- Stanley, P.E. (2005). Bioluminescence and Chemiluminescence Applications. Rapid Microbiology. A review of bioluminescent ATP techniques in rapid microbiology. Journal of Bioluminescence and Chemiluminescence 4: 375-380
- American Society for Testing of Materials (2002). D 4012-81. Volume 11.01. Standard Test Method for Adenosine Triphosphate (ATP) Content of Microorganisms in Water.
- United States Pharmacopoea 31/National Formulary 26 (2008). General Chapter <1223> ‘Validation of Alternative Microbiological Methods.
- European Pharmacopoeia 5th Edition, Supplement 5.5 (2006). 5.1.6. Alternative Methods for Control of Microbiological Quality pp. 4131-4142.







