Stabilisation of nanoparticles during freeze drying: The difference to proteins
Posted: 31 August 2011 |
The underlying concept for the stabilisation of proteins during freeze drying is the formation of a glassy matrix in which the macromolecules remain isolated and immobilised. The concept relies on the so-called ‘vitrification hypothesis’ which assumes that the formation of an amorphous phase by lyoprotectants is mandatory to interact with the amorphous protein molecule. The use of lyoprotectants has also been found to be beneficial to preserve the original particle size distribution of nanoparticles during freeze drying. Until today, it has been speculated that the predominant mechanism to suppress physical instabilities of such colloidal particle systems is their embedment in a rigid glass. Today, there are various types of colloidal particles used in drug development, and sometimes the scientific literature gives evidence that glass formation was not necessarily required for stabilisation during freezing thawing or even freeze drying. The purpose of this article is therefore to briefly provide the latest insight into potential stabilisation mechanisms when freeze drying nanoparticles, a key knowledge for rational formulation and process design for such systems.
In general, the overall goal of nanotechnology is to improve the performance of materials by the formation of particulate structures in the nanometre range1. With regard to the pharmaceutical sector, the purpose of using nanoparticles focuses on the improvement of direct interaction of an active pharmaceutical ingredient (API) with biological systems, enhancement of the stability of API’s or improvement of drug solubility2.
For that purpose, different types of pharmaceutical colloidal dosage forms (such as nanocapsules, nanosperes and drug nanoparticles) were designed over the last few years to address the respective requirements3-7. In this context, liposomal formulations can also be considered as lipid-based nanoparticulate systems8. In general, nanoparticles are physically unstable. The mechanical reduction in particle size comes with a significant increase in surface area which, in turn, is accompanied by an unfavourable increase in surface energy. Even though nanoparticles are stabilised initially during their production process (e.g. milling) in a liquid environment, particle aggregation or agglomeration and settling are frequently noticed instability issues when such systems are stored for an extended period of time9.
A possible stabilisation of colloids is by an electrostatic mechanism which implies the formation of an electrical double layer around the particles by ionic materials. The resulting coulombic repulsion counteracts the van der Waals forces as the primary source of attraction. However, electrostatic stabilisation is highly dependent on the ionic strength and quite sensitive to a shift in electrolyte concentration10. Most importantly from a pharmaceutical perspective, safety concerns regarding ionic substances must be taken into consideration if a parenteral application of such drugs is desired. For instance, cationic macromolecules readily absorb to cell membranes which could then be a major source for toxic effects11. In contrast to electrostatic stabilisation, steric stabilisation imparts colloid stability by polymers or surfactants providing a steric barrier. Such excipients can be either adsorbed through a polymeric anchor group or chemically attached to the nanoparticle surface (Figure 1)6,12-13. To overcome the attraction forces, the range of the steric barrier must be at least in excess of the van der Waals forces. Compared to electrostatic stabilisation, steric stabilisation is not very sensitive to the presence of (other) electrolytes. Further merits of this mechanism include equal efficacy at both high and low nanoparticle concentrations and the better redispersibility of flocculated particles10. However, long-term particle size stability can oftentimes not even be achieved by electrostatic, steric or electrosteric stabilisation. This fact is still a major obstacle which limits the use of liquid nanosuspensions in the pharmaceutical area. To further improve physical stability of such systems to finally achieve long-term stability, water must be removed (i.e. conversion into the solid state). Freeze drying, also referred to as lyophilisation, has already been shown as the drying technology of choice to stabilise colloidal particles14.
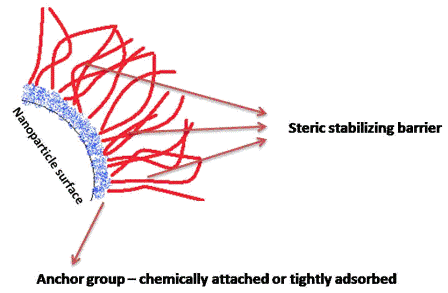

Figure 1 Schematic illustration of a particle stabilised by a steric stabiliser. Note that it has been reported that both the degree of adsorption and the density / thickness of the steric barrier are essential factors to mitigate freezing and drying stresses
Improving colloid stability by freeze drying
In particular, the parenteral administration of nanoparticles requires a maximum allowable upper particle size limit and full sterility of the suspension to avoid negative side effects like embolism and sepsis. While pharmaceutical freeze drying can be easily performed in a sterile environment, preservation of the original nanoparticle size distribution during the process is much more challenging. Freeze drying is known to impart various stresses during the individual process phases, namely (1) freezing, (2) primary drying and (3) secondary drying. All three process phases may contribute equally to an increased aggregation and agglomeration15. To protect the active compound from either freezing or drying stresses, cryoprotectants and/or lyoprotectants are typically added to such formulations. Note that many stabilisers used for freeze drying can serve as both cryoand lyoprotectants. Regardless of the type of stabiliser used, all of these materials have in common that they vitrify during freezing at the glass transition temperature of the freezeconcentrated solute, Tg’16. If glass formation is indeed necessary for stabilisation, Tg’ represents the critical formulation temperature or maximum allowable product temperature during freeze drying which must not be exceeded to avoid structural mobility. The major drawback is that freeze drying is generally known as a time consuming and therefore costly drying operation, but Tg’ values of freeze drying stabilisers are frequently found at very low temperatures. In the case that product temperatures were controlled below Tg’, the solute phase remains in an rigid amorphous glass which is compulsory to interact with a given system and therefore expected to enable stabilisation16. In the glassy state, molecular diffusion processes are minimised and the active compound is locked in so that physical instabilities are not likely to occur. However, this concept was originally developed to explain the stabilisation mechanism of proteins during freeze drying. Therefore, it has initially been assumed that this concept of stabilisation also applies for the stabilisation of nanoparticulate formulations14. This appears to be rational since it seems intuitive that suspended nanoparticles which have been arrested in a glassy solid are also immobilised, and thus isolated from each other. Surprisingly, recent literature described that typical amorphous protein stabilising lyoprotectants were not always found to be successful as protectants when freeze thawing or freeze drying colloidal formulations17,18. From a scientific point of view, it is therefore of great interest if glass formation of lyoprotectants is integral to preserve the nanoparticle size. If not, the practical consequence is that Tg’ serves not as the uppermost critical product temperature boundary during primary drying which, in turn, might allow a dramatic reduction of cycle time.
The development of the particle isolation hypothesis
The observation that some glass forming lyoprotectants are not capable of preventing physical instabilities can be explained by the particle isolation hypothesis18. The basis for this hypothesis is that the particle size is not automatically impacted when the fraction of particles is mobile in the unfrozen lyoprotectant solution. It must also be considered that stability is governed by the surface tension of the solute phase. Comparably to an emulsion, suspended droplets will only tend to coalesce in response to a high surface tension of the dispersion medium. In line with this argument, the lyoprotectant solution should prudentially possess a low surface tension, particularly at subzero temperatures as the surface tension is a function of temperature19. Allison et al. examined that hydroxethyl starch (HES) has no stabilisation capacity for colloidal, non-viral vector complexes during freezing than sucrose or glucose, despite the ability of HES to form a glassy matrix at relatively high temperatures18. Important in this discussion was that a more pronounced increase in surface tension was revealed for HES solutions during freezing, relative to sucrose or glucose solutions. In line with this, Taylor et al. observed no aggregations of colloidal nonviral vectors in an unfrozen sucrose solution above Tg’ during freezing20. Based on this finding, it was suggested that the freezing and primary drying step of lyophilisation can be performed at much higher product temperatures which would increase cycle efficiency.
The impact of the particle isolation hypothesis on freeze drying of pharmaceutical nanoparticles
From a practical perspective, the idea of increasing product temperatures above the critical formulation temperature without compromising nanoparticle size when processing a colloidal formulation is highly appealing. Thus, it is of great interest if the particle isolation hypothesis is also valid when freeze drying other types of pharmaceutical nanoparticles. Latest research indicated that Tg’ values of different used lyoprotectants and control of product temperature below this value during primary drying could not be related to particle stability of steric stabilised pure drug nanoparticles21. In this study, product temperatures were controlled either below or significantly above the corresponding Tg’ value, as determined by Differential Scanning Calorimetry. Surprisingly, the authors found comparable particle size distributions. The study could prove for the first time that the stability of the drug nanoparticles was not only a function of the physicochemical characteristics of the lyoprotectants (as proposed by the particle isolation hypothesis), but also a function of a dense and steric barrier surrounding the drug nanoparticles. Both the steric barrier and a relatively low surface tension of the unfrozen carbohydrate solution allow the drug nanoparticles sufficient mobility during primary drying. It is therefore clear that more attention must be paid on the interrelationship between the nanoparticle surface properties and corresponding dispersion medium.
In line with this argument, several authors have already emphasised the importance of polymers, such as polyvinyl alcohol (PVA) and various types of poloxamers, as steric stabilisers to reduce freezing drying stress. However, the role of the physicochemical state of such materials during freeze drying has not been investigated so far22,23. The concept of the formation of a thick layer on top of the nanoparticle surface which improves the resistance to physical instability was successfully applied many times. It has even been successfully demonstrated that freeze drying of nanoparticles can be performed without the addition of any freeze drying stabiliser which required only the choice of an adequate steric stabiliser23,24. Moreover, it has been published recently that a minimum molecular weight, i.e. chain length, of the steric stabiliser and certain degree of adsorption of the stabiliser on the nanoparticle surface is essential to prevent freezing and drying stress (Figure 1, page 12). This observation served as another hint that glass formation is not the dominant stabilisation mechanism when freeze drying nanoparticles23.
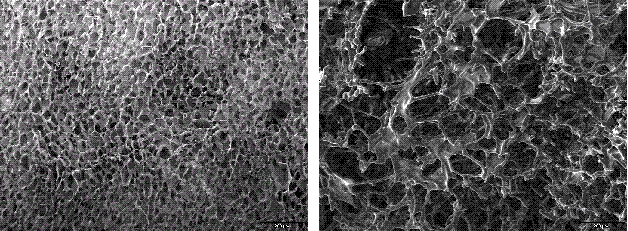

Figure 2 SEM pictures of the inner cake morphology of lyophilised sucrose solutions when processed either above (left) and below (right) Toc. Maintaining the product temperature above Toc during primary drying resulted in viscous flow of the porous excipient matrix
The maximum allowable product temperature when freeze drying nanosuspensions
As Tg’ does not perfectly reflect the maximum allowable product temperature during primary drying in real freeze drying cycles, the onset of collapse temperature, Toc, is routinely used as a more representative measure of the critical formulation temperature for freeze drying25,26. This temperature is obtained from Freeze Dry Microscopy, an analytical procedure which realises freeze drying of a formulation in microscale. As a rule of thumb, Toc is approximately 2°C to 5°C higher than the corresponding Tg’ value25. Maintaining the product temperature above Toc induces first changes in the inner cake morphology due to viscous flow of the (dried) structure (Figure 2). As a consequence, macroscopic cake defects (cake shrinkage or collapse) might be observed in the final product which compromise product elegancy. Importantly, it has been reported that such cake defects also influence pharmaceutically relevant performance attributes, such as product reconstitution times, completeness of reconstitution, API stability, residual moisture, and more25,26. Interestingly, it has been found that a high nanoparticle concentration might provide additional cake stability when freeze drying nanosuspensions21. It was argued that the high total solid content of suspended drugnanoparticles counteracts structural deterioration as shown in Figure 3. Moreover, formulations containing a lower lyoprotectant concentration revealed neither shrinkage nor microcollapse, even though product temperature clearly exceeded the onset of collapse temperature. As a consequence, the product temperature during primary drying may be optimised in the case that the formulation design is optimised. In this case, not only would the formulation be the limitation for the process, but the freeze drying equipment design will impose the restrictions to the cycle conditions.
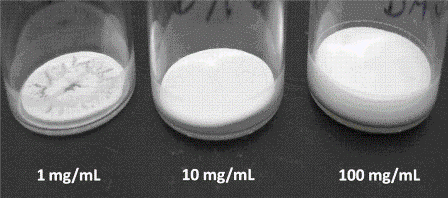

Figure 3 Cake appearance as a function of nanoparticle concentration after freeze drying of pure drug nanosuspensions well above the investigated critical formulation temperatures and without any addition of freeze-drying stabilising agents
Conclusions
Immobilisation of the API by formation of a rigid glass is an effective mechanism to prevent physical instabilities during freeze drying. However, in comparison to freeze drying of proteins nanoparticles do not necessarily require a glassy lyoprotectant matrix in which the colloidal fraction is confined and isolated from each other. According to the particle isolation hypothesis, nanoparticles are allowed to have freedom of mobility in the unfrozen lyoprotectant solution which means that they remain well dispersed throughout the process. A preferably low surface tension of the excipient solution is more important to prevent physical instabilities during freeze drying than glass formation of lyoprotectants at relatively high subzero temperatures. Further, if the colloidal materials are already pre-stabilised in a liquid environment prior to freeze drying, specific nanoparticle stabilising materials (e.g. steric stabilisers) can clearly improve their resistance to stresses imposed by the freezing and drying step of a freeze drying cycle. Lastly, latest research has proved that freeze drying time can be greatly shortened as product temperature can be increased well above the critical formulation temperature of the nanoparticle formulation.
References
- The Royal Society. Nanoscience and nano – technologies: opportunities and uncertainties. London: Royal Society, 2004. ISBN: 0 85403 604 0
- D. Thassu, Y. Pathak, M. Deleers. Nanoparticulate Drug- Delivery Systems: An Overview, in: D. Thassu, Y. Pathak, M. Deleers (Eds.). Nanoparticulate Drug- Delivery Systems, informa healthcare, New York, 2007, pp. 1-31
- S.R. Schaffazick, A.R. Pollmann, T. Dalla-Costa, S.S. Guterres. Freeze-drying polymeric colloidal suspensions: nanocapsules, nanospheres and nanodispersion. A comparative study. Eur. J. Pharm. Biopharm. 56 (2003) 501-506
- R. Oppenheim. Solid colloidal drug delivery systems: nanoparticles. Int. J. Pharm. 8 (1981) 217-234
- M.J. Alonso. Nanoparticulate drug carrier technology, in: S. Cohen, H. Bernstein (Eds.). Microparticulate Systems for the Delivery of Proteins and Vaccines, Marcel Dekker, New York, 1996, pp. 203-242
- E. Merisko-Liversidge, G.G. Liversidge, E. Cooper. Nanosizing: A formulation approach for poorly-watersoluble compounds. Eur. J. Pharm. Sci. (2003) 18:113
- E. Merisko-Liversidge. Nanocrystals:resolving pharmaceutical formulation issues associated with poorly water-soluble compounds in particles. Orlando: Marcel Dekker, 2002
- J. Wu, X. Zhao, R.J. Lee. Lipid-Based Nanoparticulate Drug Delivery Systems, in: D. Thassu, Y. Pathak, M. Deleers (Eds.). Nanoparticulate Drug-Delivery Systems, informa healthcare, New York, 2007, pp. 89-98
- T. Cosgrove (Ed.). Colloid Science – Principles, methods and applications. Blackwell Publishing, 2005. ISBN: 1 4051 2673 6
- D. H. Napper. Polymeric Stabilization of Colloidal Dispersions. Academic Press, London, 1983
- P. Brummer. Interfacial phenomena, in: A. Gennaro (Ed.), Remington: The Science and Practice of Pharmacy, 20th Ed., Lippincott Williams & Wilkins, Baltimore, 2000, p. 285
- F. Kesisoglu, S. Panmai, Y. Wu. Nanosizing – Oral formulation development and biopharmaceutical evaluation. Adv. Drug Del. Rev. (2007) 56: 631-644
- C. Lourenco, M. Teixeira, S. Simoes, R. Gaspar. Steric stabilization of nanoparticles: Size and surface properties. Int. J. Pharm. (1996) 138: 1-12
- W. Abdelwahed, G. Degobert, S. Stainmesse, H. Fessi. Freeze-drying of nanoparticles: Formulation, process and storage considerations. Adv. Drug Deliv. Rev. 58 (2006) 15:1688-1713
- M.J. Pikal. Freeze-Drying, in: J. Swarbrick (Ed.), Encyclopedia of Pharmaceutical Technology, vol.2, Marcel Dekker, New York, 2002, pp. 1299-1326
- W. Wang. Lyophilization and development of solid protein pharmaceuticals. Int. J. Pharm., 203 (2000), pp. 1-60
- J. Beirowski, S. Inghelbrecht, A. Arien, H. Gieseler. Freeze drying of nanosuspensions, 1: Freezing rate versus formulation design as critical factors to preserve the original particle size distribution. J. Pharm. Sci. (2011) 100 (5): 1958-1968
- S.D. Allison, M.D.C. Molina, T.J. Anchordoquy. Stabilization of lipid/DNA complexes during the freezing step of the lyophilization process: The particle isolation hypothesis. Biochim. Biophys. Acta. (2000) 1468: 127-138
- K.J. Laidler, J.H. Meiser, Physical Chemistry, Benjamin/Cummings, Menlo Park, CA, 1982.
- T.K. Armstrong, T.J. Anchordoquy. Immobilization of nonviral vectors during the freezing step of lyophilisation. J. Pharm. Sci. (2004) 93: 2698-2709
- J. Beirowski, S. Inghelbrecht, A. Arien, H. Gieseler. Freeze drying of nanosuspensions, 2: The role of the critical formulation temperature on stability of drug nanosuspensions and its practical implication on process design. J. Pharm. Sci. (2011) accepted
- H. Takeuchi, H. Yamamato, T. Toyoda, H. Toyobuku, T. Hino, Y. Kawashima. Physical stability of size controlled small unilamellar liposomes coated with a modified polyvinyl alcohol. Int. J. Pharm. (1998) 164:103-111
- J. Beirowski, H. Gieseler, S. Inghelbrecht, T. Arien, I. Van Assche. Investigation of various poloxamers as freeze-drying stabilizers in freeze-drying of nanosuspensions. Proc. 2009 PDA Pharmaceutical Freeze Drying Technology; Sep. 29-30 (2009); Frankfurt, Germany
- F. de Jaeghere, E. Allemann, J.C. Leroux, W. Stevels, J. Feijen, E. Doelker, R. Gurny. Formulation and lyoprotection of poly (Lactic acid-co-ehtylene oxide) nanoparticles: influence on physical stability and in vitro cell uptake. Pharm. Res. (1999) 16: 859-866
- M.J. Pikal, S. Shah. The collapse temperature in freezedrying: Dependence on measurement methodology and rate of water removal from the glassy phase. Int. J. Pharm. (1990) 62: 165-186
- E. Meister, H. Gieseler. Freeze-dry microscopy of protein/sugar mixtures: Drying behavior, interpretation of collapse temperatures and a comparison to corresponding glass transition data. J. Pharm. Sci. (2009) 98 (9): 3072-3087








