Is the compendia-recommended microorganism test panel truly representative?
Posted: 23 April 2021 | Hannah Balfour (European Pharmaceutical Review) | 3 comments
A recently published paper suggests the test panel of microorganisms needs to be reviewed and likely expanded to ensure it is suitable for application in the pharma industry.
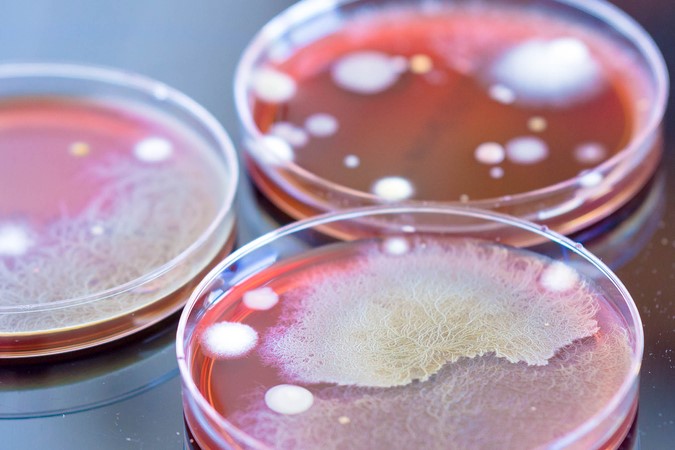

In a recent paper published in EJPPS,1 Dr Tim Sandle argued that the test panel of microorganisms currently recommended by the major pharmacopoeia for microbiological method suitability studies, disinfectant efficacy studies, antimicrobial effectiveness testing and associated activities may not be suitable for their intended application.
In the paper, Dr Sandle explained that because the pharmacopoeia microbial panel recommendations are designed to be multi-industry, are often applied for a broader range of activities or tests than specified and have failed to keep up with the industry’s understanding of the types of microorganisms likely to cause contamination, they may not be representative of what could be recovered.
He added that this is especially true in cleanroom environmental monitoring, where the selection of microorganisms for the test panel has not adapted to incorporate the findings from the human microbiome, which explored the depth of species richness found on the human skin microbiome.
Instead of the current compendia-recommended panel, which Dr Sandle said is only representative of what microorganisms may be recovered if you are concerned with the recovery of non-fastidious mesophilic organisms, he recommends that the panel should be based on what is currently being recovered and should theoretically be recovered.
To achieve the necessary representation, Dr Sandle recommend that the test panel of challenge organisms be widened, for instance to include Cladosporium species in the panel for cleanroom monitoring, since they are one of the most commonly isolated fungi. Additionally, he suggested Ralstonia pickettii be included in the purified water system test panel, since it is the most common isolate recovered there, and that Staphylococcus aureus, carried in the nostrils of almost a quarter of the population should be augmented by organisms such as Micrococcus luteus or Staphylococcus epidermidis, carried in greater abundance in the skin.
He added that to decide what should be included in their testing panel, microbiologists should regularly screen and trend the microorganisms recovered according to area monitored and sample type. This, said Dr Sandle, can also assist with contamination control.
In the paper Dr Sandle also explores whether replacement organisms or panel extensions should be made up of cultures purchased from a culture collection, which would permit inter-laboratory reproducibility, or made up of wildtype (environmental) isolates from the manufacturing site, since they are more representative of the specific facility’s microbiota.
He stated that arguments can be made for either, because while environmental isolates may be more representative of the facility, they are not standardised and can sometimes be challenging to recover due to the conditions they may have been exposed to (eg, disinfectants etc). Alternatively, cultures derived from culture collections have resided on nutritive rich media over many generations have adapted for this kind of recovery, so may not be sufficient to provide confidence that the medium can recover sub-lethally damaged organisms, that may have been exposed to stressors in cleanroom conditions, such as disinfectant.
Overall, Dr Sandle stated that the test panel of microorganisms should be reviewed and likely expanded, with some or all of the panel to be replaced and varied based on application. As such, he said that the size of the panel would vary, since it needs to be based on risk assessment and balance practicalities and resources against representativeness.
Reference
- Sandle T. Why it’s time to strengthen and widen the microbial test panel. EJPPS EUROPEAN JOURNAL OF PARENTERAL AND PHARMACEUTICAL SCIENCES [Internet]. 2021 [cited 23 April 2021]. Available from: https://www.ejpps.online/strengthen-widen-microbial-panel
Related topics
Cleanrooms, Drug Manufacturing, Drug Safety, Microbial Detection, Microbiology, Microbiomes, QA/QC










USP states “Challenge organisms are generally based on likely contaminants to a drug product while considering its physical attributes, formulation, and intended use. The standard battery of challenge organisms described in this test need not prevent the inclusion of other species of microorganisms if deemed useful to measure the biological activity of the preservative system for a specific product. These supplemental challenge organisms are not within the scope of this chapter, but may be added in addition to the described test organisms.”
The test panel used by the compendia was chosen based on the most aggressive species in each group. It was designed to test against a representative sample not every possible recovery.
It is the site’s responsibility to add native organism to any test method or disinfectant efficacy evaluation for robustness.
Very interesting, this makes sense and give smuch to think about in the lab.