EC grants marketing authorisation of Dupixent for atopic dermatitis
Posted: 28 September 2017 | Dr Zara Kassam (European Pharmaceutical Review) | No comments yet
Sanofi and Regeneron Pharmaceuticals, Inc. have announced that the EC has granted marketing authorisation for Dupixent for use in adults with moderate-to-severe atopic dermatitis…
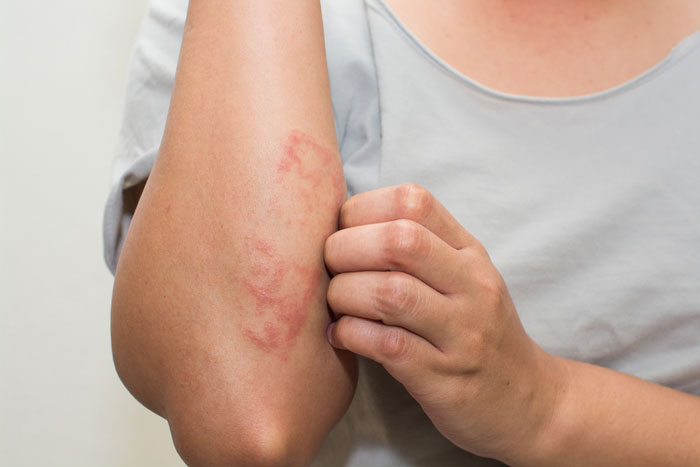

Sanofi and Regeneron Pharmaceuticals, Inc. have announced that the European Commission has granted marketing authorisation for Dupixent (dupilumab), for use in adults with moderate-to-severe atopic dermatitis (AD) who are candidates for systemic therapy.
“People with moderate-to-severe atopic dermatitis can experience unbearable symptoms that may significantly impact their quality of life. Many often struggle to control their disease with the treatment options currently available,” said Christine Janus, Chief Executive Officer of the International Alliance of Dermatology Patient Organizations.
“We support timely access to this important new medication for those with moderate-to-severe atopic dermatitis to help them control and provide relief for this life-altering, often severely debilitating, chronic disease,” she added.
Dupixent is a human monoclonal antibody that is designed to specifically inhibit overactive signalling of two key proteins, IL-4 and IL-13, which are believed to be major drivers of the persistent underlying inflammation in atopic dermatitis, and certain other allergic or atopic diseases.
Dupixent will come in a pre-filled syringe and can be self-administered by a patient as a subcutaneous injection every other week after an initial loading dose. Dupixent can be used with or without topical corticosteroids.
“This approval of Dupixent in Europe demonstrates our approach of bringing innovative new therapies to those living with high unmet medical need and today’s approval represents an important milestone for people living with moderate-to-severe atopic dermatitis in Europe,” said Dr Elias Zerhouni, President, Global R&D, Sanofi.
“Dupixent targets an underlying cause of atopic dermatitis helps clear the skin, manage the persistent debilitating itch and improve the overall quality of life. We are now focused on quickly making this important new treatment option available to people across Europe who live with this systemic disease,” Dr Zerhouni added.
Following the granting of this marketing authorisation, Sanofi and Regeneron will work with relevant local authorities to make Dupixent available to patients in need in countries across Europe.
“Dupixent represents the culmination of decades of our scientific research into the biology of allergic diseases such as moderate-to-severe atopic dermatitis,” said Dr George D. Yancopoulos, President, and Chief Scientific Officer, Regeneron. “We continue to evaluate the potential of dupilumab in the treatment of atopic dermatitis in children and adolescents as well as other allergic inflammatory diseases driven by the IL-4/IL-13 pathway.”
Dupixent is approved in the U.S. for the treatment of adults with moderate-to-severe atopic dermatitis that is not well controlled with prescription therapies used on the skin, or who cannot use topical therapies. Dupixent can be used with or without topical corticosteroids. It is not known if Dupixent is safe and effective in children.
Dupixent can be used with or without topical corticosteroids. It is not known if Dupixent is safe and effective in children.
Atopic dermatitis, a form of eczema, is a chronic inflammatory disease with symptoms often appearing as a rash on the skin. Moderate-to-severe atopic dermatitis is characterized by rashes often covering much of the body, and can include intense, persistent itching and skin dryness, cracking, redness, crusting, and oozing. Itch is one of the most burdensome symptoms for patients and can be debilitating. In addition, people with moderate-to-severe atopic dermatitis experience impaired quality of life, including disrupted sleep, and increased anxiety and depression symptoms along with their disease.
Related topics
Related organisations
European Commission, nternational Alliance of Dermatology Patient Organizations, Regeneron Pharmaceuticals, Sanofi
Related drugs
Related people
Christine Janus, Dr George D. Yancopoulos, Dr. Elias Zerhouni