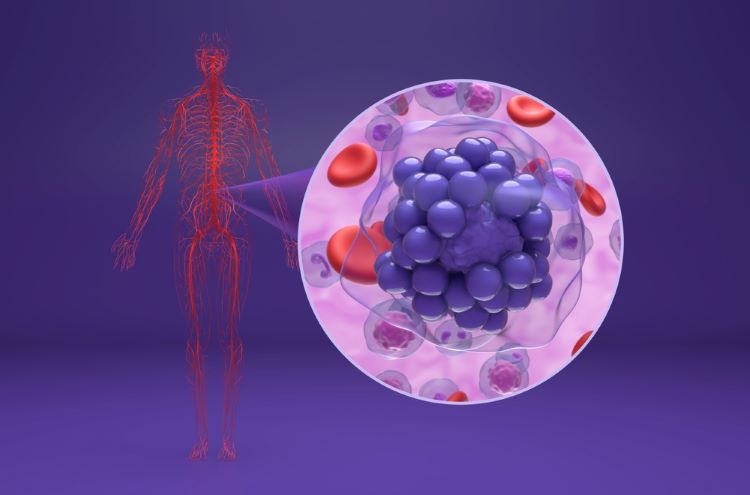
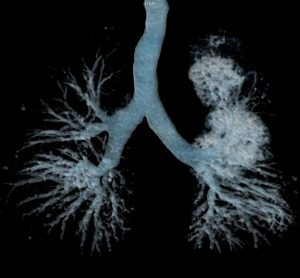
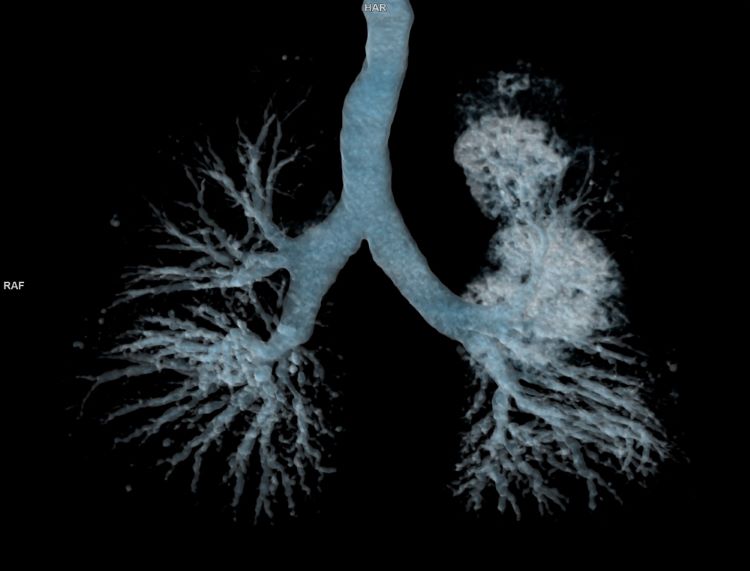
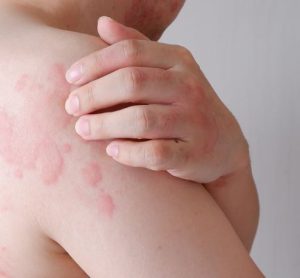
Innovative CAR T therapy could deliver in broader therapeutic settings, research suggests
The novel cell therapy utilises a shortened manufacturing process compared to standard production timelines, offering potential for enhanced therapeutic capability.