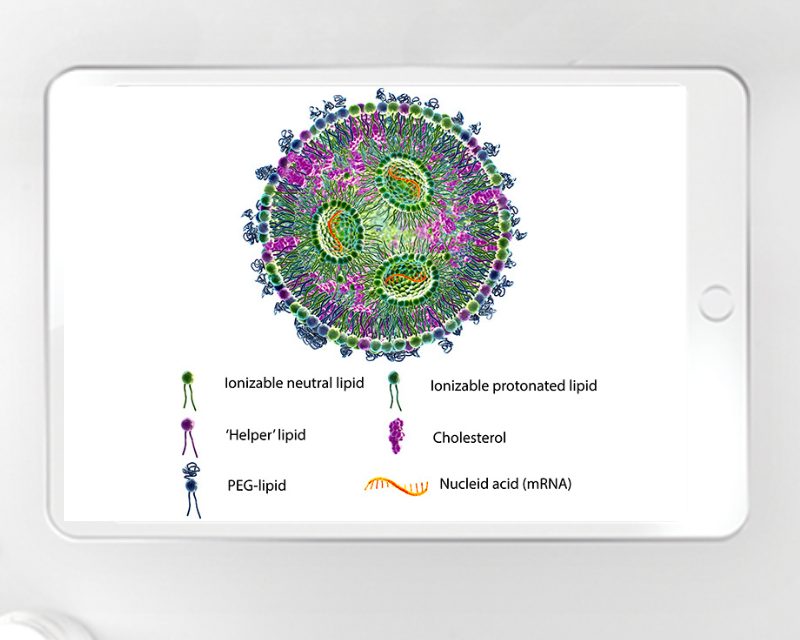
Breaking down silos – how digital tools can transform pharma manufacturing
Following their recent LinkedIn Live, Hexagon’s Adam Cross, Industry Director for Pharma and Life Sciences, reveals the state of data in pharmaceutical manufacturing, the opportunities available and how companies can ensure success and sustainable outcomes in their digitalisation journey.