How to improve vaccine procurement strategies to optimise coverage in Europe
Posted: 8 June 2020 | Ryan Lawlor (Charles River Associates), Tim Wilsdon (Charles River Associates) | 1 comment
Tim Wilsdon and Ryan Lawlor from Charles River Associates explain why price-based procurement strategies can limit the long-term effectiveness and sustainability of vaccine programmes.

Vaccination programmes implemented across Europe and other regions around the world play a central role in the prevention and spread of many serious infectious diseases and have brought significant benefits to both patients and clinicians. These programmes are estimated to save the lives of more than 2.5 million children worldwide each year and are responsible for the eradication of smallpox as well as the elimination of measles, diphtheria and rubella in many regions.1,2 With the ongoing COVID-19 pandemic and the global focus on developing a vaccine, the importance of government-sponsored vaccination programmes is clearer than ever.
There are a variety of models that are used to procure vaccines for large-scale immunisation programmes, governed by the European legal framework for procurement. These range from price-based models, designed to acquire vaccines at the lowest cost possible, to value-based models that focus on a drug’s perceived value to clinicians, patients and healthcare systems. Price-based models are popular in many countries as they result in lower costs in the short term. However, a recent analysis led by the Life Sciences team at Charles River Associates (CRA) indicates that a price-based vaccine procurement model can limit the long-term effectiveness and sustainability of many vaccination programmes and potentially put public health at risk.
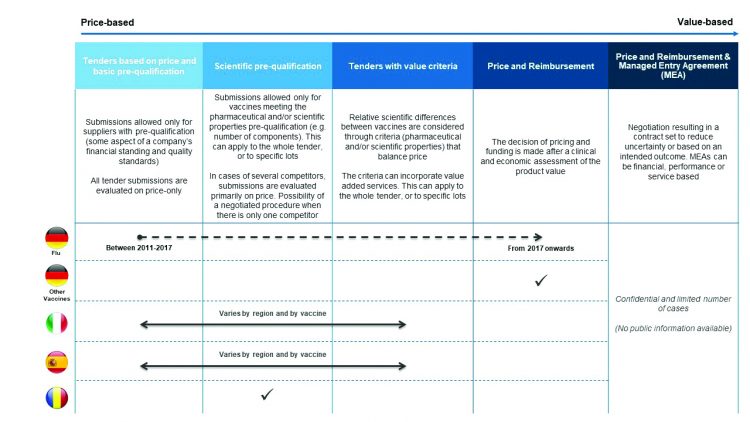

Figure 1: Types of procurement mechanisms for vaccines [credit: CRA analysis].
To better understand how vaccine procurement mechanisms can impact the overall sustainability of immunisation programmes and the ability to meet public health goals, the team reviewed programme descriptions and assessments from multiple sources including government websites, databases and peer-reviewed journal publications. This was complemented by interviews with a range of stakeholders in the life sciences sector including government officials, academics, payers and public health specialists. The focus was placed on countries that use a range of procurement mechanisms, from those which primarily focus on price to those that take a more value-based approach (Figure 1). Germany, Italy, Spain and Romania were selected as countries with different mechanisms in place. The relationship was then documented between procurement model dynamics and the level of protection against vaccine preventable diseases: measles, hexavalent and influenza vaccines.
Challenges with price-based procurement
Research shows that the use of price-based models, particularly those involving a ‘winner-takes-all’ approach, will typically result in often significant short-term reductions in vaccine costs. In one example, when Germany first introduced price-based procurement for influenza vaccines in 2011 the price subsequently dropped by about 30 percent in the first year and by about 56 percent the following year (Figure 2). In contrast, prices for vaccines that were not purchased based on a lowest-cost model – including those for hexavalent and measles, mumps and rubella (MMR) – remained consistent during the same period.
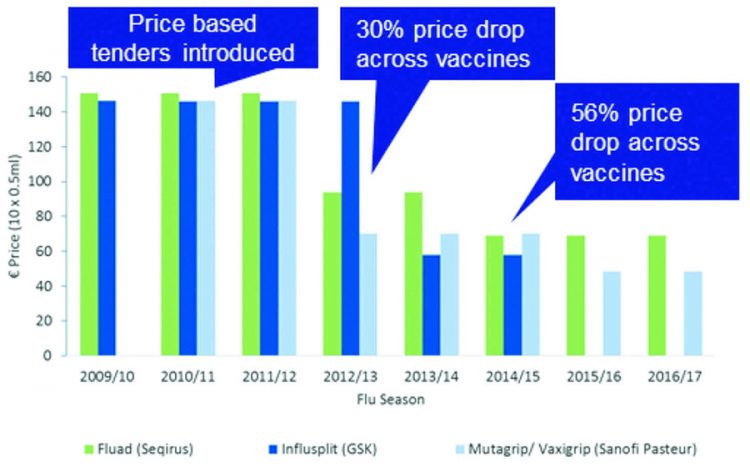

Figure 2: Germany – influenza vaccine prices 2009–2017 (10×0.5ml) [credit: Lauer-Taxe].
Although price-based models can bring short-term financial savings, they may be associated with reduced vaccination coverage rates (VCRs) in Europe. Routine immunisation coverage of the first dose of a measles-containing vaccine has declined in 12 EU states since 2010, accompanied by a resurgence in measles cases during the same period.3,4 In Spain, recent efforts to achieve lower vaccine prices in all regions, following a national framework agreement implemented in 2011, have correlated with a decline in influenza VCRs (Figure 3). While this did result in a 37 percent price reduction, there was also evidence of a 14 percent fall in VCR. Similarly, analysis of trends in both the Puglia region of Italy and Romania found a decline in VCRs during a period when vaccine prices also were reduced. Overall, evidence shows that lower prices have not been able to maintain or increase VCR across countries.
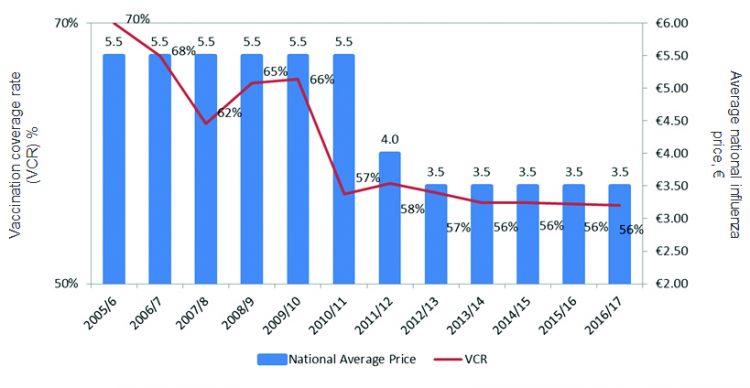

Figure 3: Influenza vaccine price and vaccine coverage rate in Spain (2005–2017) [credit: Ministry of Health, Social Services and Equality].
Price-based models have also been found to exacerbate supply issues. For example, since Spain began purchasing vaccines based on price, the country has seen difficulty identifying suppliers – particularly for diphtheria, MMR, tetanus, pertussis, pneumococcus, typhoid fever, rabies and yellow fever vaccines, due to the lower commercial attractiveness of the terms.5 In response, government officials increased the MMR vaccine price in 2014 to incentivise manufacturers to continue supplying the Spanish market.6 When manufacturers find a market less attractive and decline to participate, this can increase the potential for supply interruptions and reduced competition. In the long term, the impact on market competition and manufacturers’ willingness to invest in R&D could stagnate innovation, leading to areas of unmet need.
…price-based vaccine procurement model can limit the long-term effectiveness and sustainability of many vaccination programmes”
The complex and lengthy production process required for vaccines, including many that are combination therapies, is also an important consideration that can potentially affect supply. In 2012, Germany awarded a sole supplier the responsibility of producing influenza vaccines for several states. When the supplier was unable to deliver the one million doses it promised due to identified impurities, their exclusive contract meant that there were no other influenza vaccine suppliers with available stock.7 Manufacturers often cannot rapidly increase production or reallocate vaccine doses even if they are positioned as ‘back-up’ suppliers because of late demand and necessary production timelines. Poor planning of local supply needs and manufacturing timeframes can lead to vaccine shortages and failure to dose populations within appropriate immunisation windows, potentially putting thousands of people at risk of developing otherwise preventable diseases.
Innovative strategies in vaccine procurement

Some countries are also transitioning to procurement mechanisms that prioritise value-added services in an effort to increase or maintain VCRs. The Spanish and Italian governments are increasingly realising that anti-vaccination campaigns and a lack of awareness and understanding of vaccine public health benefits are significantly impacting VCRs. As a result, some regions are requiring vaccine purchasing agreements to include value-based services to increase the availability of educational resources for clinicians and patients.
Strategies moving forward
Based on these analyses, there is compelling evidence indicating that procurement strategies based solely or primarily on price can put the future sustainability of global vaccination programmes at risk. These strategies are shown to increase the risk of supply shortages and limit public understanding of the health benefits of vaccines. Furthermore, the addition of value-added services such as training programmes for healthcare professionals, public awareness campaigns, better patient monitoring and vaccines registries can play an important role in improving outcomes and supporting long-term sustainability.
…some regions are requiring vaccine purchasing agreements to include value-based services”
The need for more innovation in vaccine procurement strategies is also driven by recent recommendations from the EU Council calling for countries to strengthen cross-border co-operation in the fight against vaccine-preventable diseases. These recommendations encourage EU member states and the European Commission (EC) to develop joint actions aimed at sharing best practices on vaccination policies.10 It will be important for manufacturers and purchasers to communicate openly and collaborate to address challenges together. Consistent engagement will allow both parties to align on solutions that can help ensure the long-term sustainability of vaccination programmes and improve the health and quality of life of many people around the world in the years ahead.
About the authors




The authors wish to acknowledge the contributions of Alexandru Rafila, Carol Davila University of Medicine and Pharmacy; Amós García Rojas, Epidemiology and Prevention Servicio Canario de la Salud to this article. Funding for this project came from Sanofi Pasteur.
The views expressed herein are the authors’ and not those of Charles River Associates (CRA) or any of the organisations with which the authors are affiliated.
References
- A. Orenstein, R. Ahmed. Simply put: Vaccination saves lives. PNAS. 2017 Apr 18; 114 (16): 4031-4033. Available from: http://www.pnas.org/content/114/16/4031
- International Federation of Pharmaceutical Manufacturers & Associations. The Pharmaceutical Industry and Global Health. Facts and Figures 2017 [Internet]. International Federation of Pharmaceutical Manufacturers & Associations; 2017 [cited 2017 Sept 12]. Available from: https://www.ifpma.org/wp-content/uploads/2017/02/IFPMA-Facts-And-Figures-2017.pdf
- Larson, A. de Figueiredo, E. Karafillakis, M. Rawal. State of vaccine confidence in the EU 2018 [Internet]. Luxembourg: Publications Office of the European Union; 2018 [cited 2019 Sept 18]. Available from: https://ec.europa.eu/health/sites/health/files/vaccination/docs/2018_vaccine_confidence_en. pdf DOI: 10.2875/241099
- World Health Organization. Global and Regional Immunization Profile: European Region [Internet]. World Health Organization; 2018 [cited 2019 Sept 18]. Available from: http://www.who.int/immunization/monitoring_surveillance/data/gs_eurprofile.pdf?ua=1
- Fernandez. Tétanos, papiloma, hepatitis, neumococo, difteria … Madrid se queda sin vacunas. [Internet]. El Confidencial; 2017 Mar 3 [cited 2020 Mar 12]. Available from: https://www.elconfidencial.com/espana/madrid/2017-03-03/vacunas-madrid-escasez-desabastecimiento-tetanos-papiloma-difteria_1341589/
- P. Vincente. Es sostenible el modelo actual de aquisicion de vacunas para el calendario infantile de vacunacion? [Internet] Economía de Salud; 2016 Oct; 11 [cited 2017 Aug 15]. Available from: http://www.economiadelasalud.com/pdf/V11N4/Sostenible_modelo_calendario_infantil_vacunacion.pdf
- Stockrahm. Keine Exklusivverträge mit Pharmakonzernen! [Internet] Zeit Online; 2012 Oct 26 [cited 2017 Aug 15]. Available from: http://www.zeit.de/wissen/gesundheit/2012-10/grippe-impfstoff-novartis-engpass-kommentar
- Monitorul Oficial al Romaniei. LEGE Nr. 98/2016 din 19 ma 2016 privind achizitiile publice. [cited 2019 Sept 12]. Available from: http://www.mmuncii.ro/j33/images/Documente/Legislatie/Assistenta-sociala-2018/Legea_98_2016_la_18012018.pdf
- M. Danzon, N. S. Pereira. Vaccine Supply: Effects Of Regulation And Competition. Int J Econ Bus. 2011 July; 18(2): 239-271. Available from: http://www.nber.org/papers/w17205 DOI: 10.3386/w17205 DOI: 10.3386/w17205
- Council recommendation of 7 December 2018 on strengthened cooperation against vaccine-preventable diseases. EUR-Lex; 2018 [cited 2019 Sept 12]. Available from: https://eur-lex.europa.eu/legal-content/GA/TXT/?uri=OJ:JOC_2018_466_R_0001
Related topics
Drug Markets, Industry Insight, Regulation & Legislation, Vaccines
Related organisations
Related diseases & conditions
Covid-19, diphtheria, hexavalent, Influenza, measles, mumps and rubella (MMR), pertussis, pneumococcus, rabies, rubella, Smallpox, tetanus, typhoid fever, Yellow Fever










What a fun read – I’ll be sharing with my husband. This is appreciated!