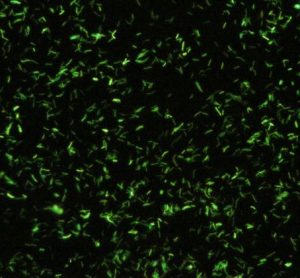
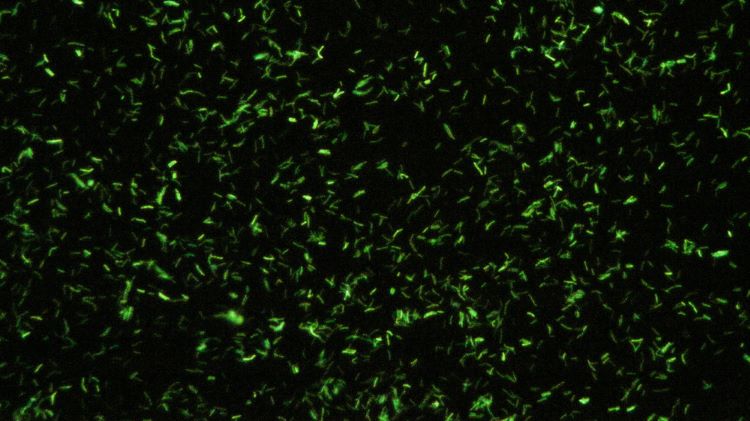
Innovative method could address a major limitation in fluorescence microscopy
With fluorescence imaging an essential tool in noninvasive biomedical research, the microscopy advancement holds opportunity for progress in optical imaging, according to the paper.