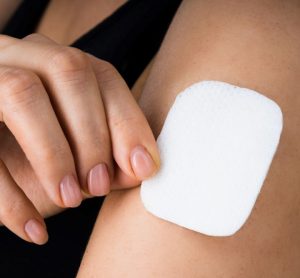
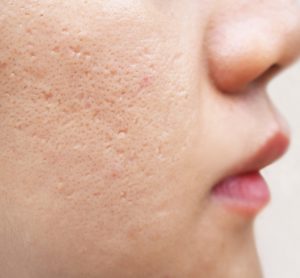
Microneedle patch with precise vancomycin drug dose against MRSA skin infections
Effective vancomycin concentrations can be delivered locally to methicillin-resistant Staphylococcus aureus (MRSA)-infected sites of the skin with the use of miniaturised needles. This presents opportunities for targeted delivery of medication for focal skin diseases. Here, Georgios A Sotiriou and Liv Eidsmo of the Karolinska Institutet discuss some of them.