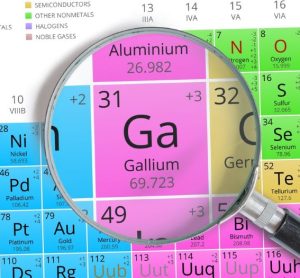
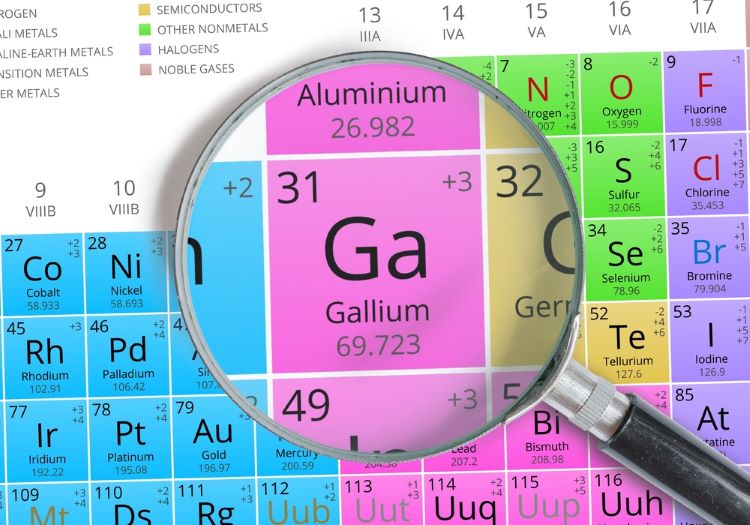
New partnership to aid supply of Ga-68-based radiopharmaceuticals
The new collaboration is set to ensure a steady, more accessible supply of Ga-68-based radiopharmaceuticals, thereby helping to address traditional logistical challenges due to gallium’s short half-life.

























![Roche logo sign lit up [Credit: testing/Shutterstock.com].](https://www.europeanpharmaceuticalreview.com/wp-content/uploads/Roche-3-300x278.jpg)
![Roche logo sign lit up [Credit: testing/Shutterstock.com].](https://www.europeanpharmaceuticalreview.com/wp-content/uploads/Roche-3-e1652444508201.jpg)



