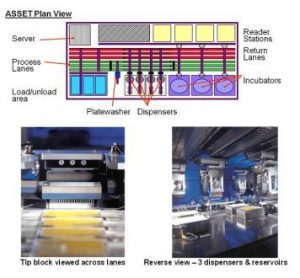
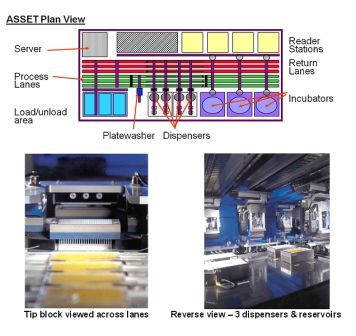
Analytical methods used in obtaining higher order structure information from protein therapeutics
The efficacy, safety and pharmacokinetic properties of a protein therapeutic substantially depend on the molecule having the right structure. This article reviews current methods used for obtaining higher order structure information of biotherapeutics.