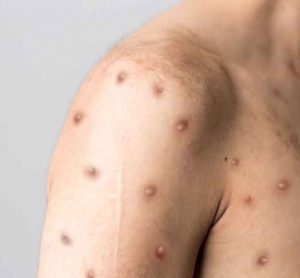
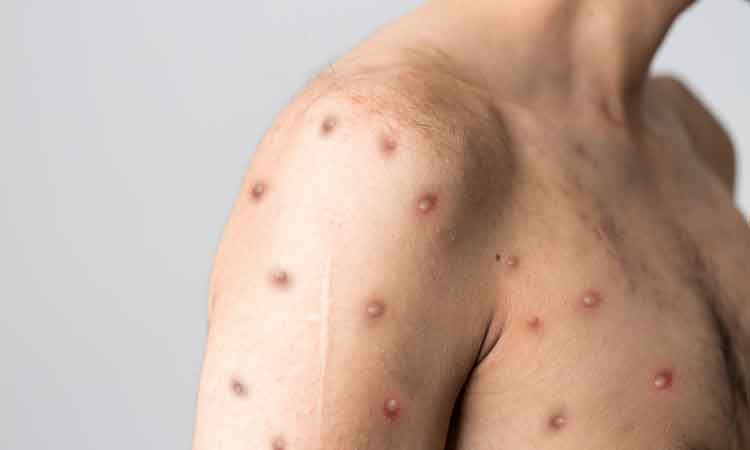
Mpox update – August 2024
This article discusses the impact of the recent mpox emergency following the outbreak of the virus in 2022, highlights the industry’s response, potential vaccine candidates and key recommendations for international management of the disease.