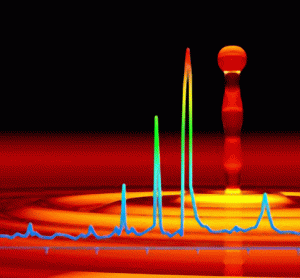
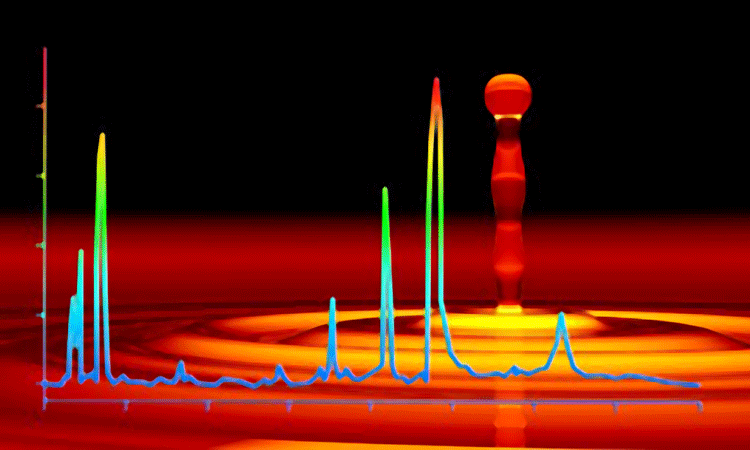
The obstacle to developing an optimum placebo formulation for pharmaceutical freeze drying
Scale-up of pharmaceutical freeze-drying processes is cost and time consuming due to Quality by Design (QbD) requirements and novel compound costs or availability. The application of placebo formulations offers a promising way to improve drug development activities and save resources. Furthermore, placebos are used as a reference in clinical trials.…