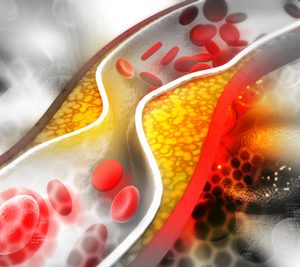
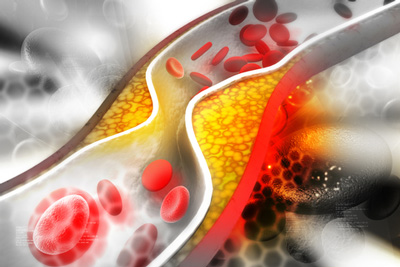
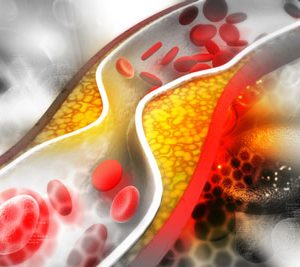
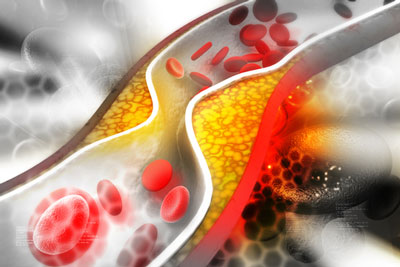
siRNA therapy shows long-term potential in lowering LDL-C
New data presented at the European Society of Cardiology (ESC) Congress 2023, showed Novartis’ Leqvio® (inclisiran) provided an approximate 49 percent reduction in LDL cholesterol beyond six years of clinical observation.







![MIT chemical engineers have devised a new way to formulate hydrophobic drugs by turning them into a nanoemulsion, which can be poured into a mold and dried into tablets. The nanoemulsion can also be used to form small particles, seen in the vials [Credit: Felice Frankel].](https://www.europeanpharmaceuticalreview.com/wp-content/uploads/MIT-HYDROPHOBIC-DRUGS-nanoemulsion-300x278.jpg)
![MIT chemical engineers have devised a new way to formulate hydrophobic drugs by turning them into a nanoemulsion, which can be poured into a mold and dried into tablets. The nanoemulsion can also be used to form small particles, seen in the vials [Credit: Felice Frankel].](https://www.europeanpharmaceuticalreview.com/wp-content/uploads/MIT-HYDROPHOBIC-DRUGS-nanoemulsion-e1623151984970.jpg)