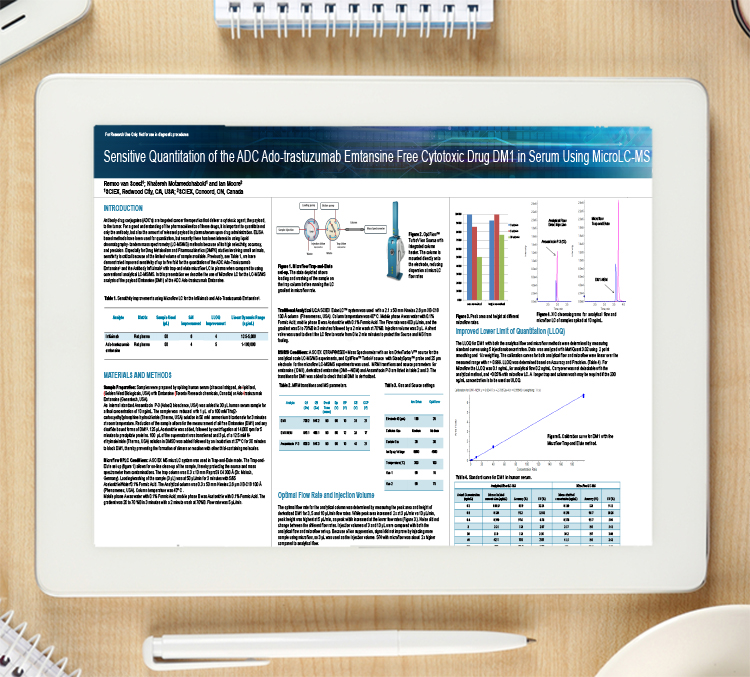
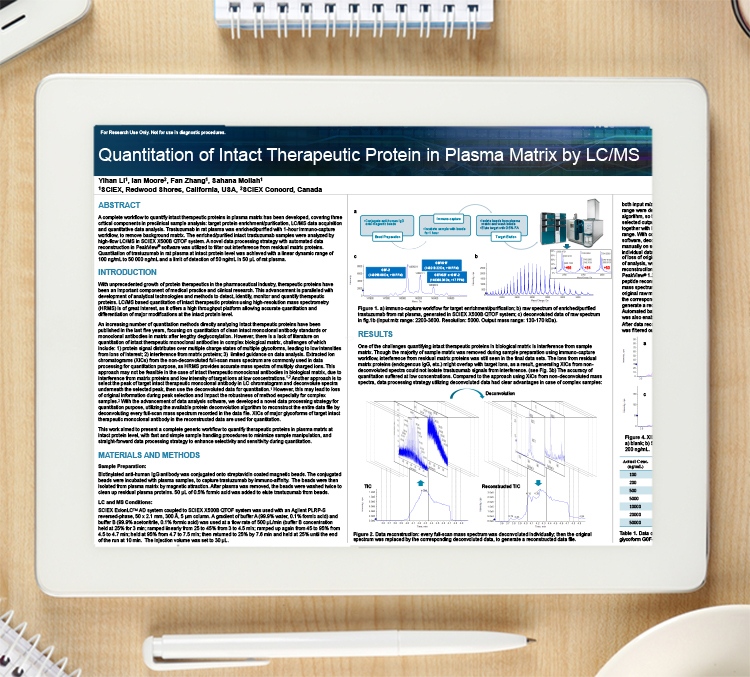
Application note: Improving the efficiency of catalyst screening in drug substance development
Herewith ACD/Labs presents their web-based software solution to the multiple challenges experienced in screening workflows; offering effective support for high-throughput catalyst screening.