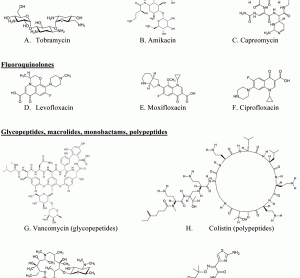
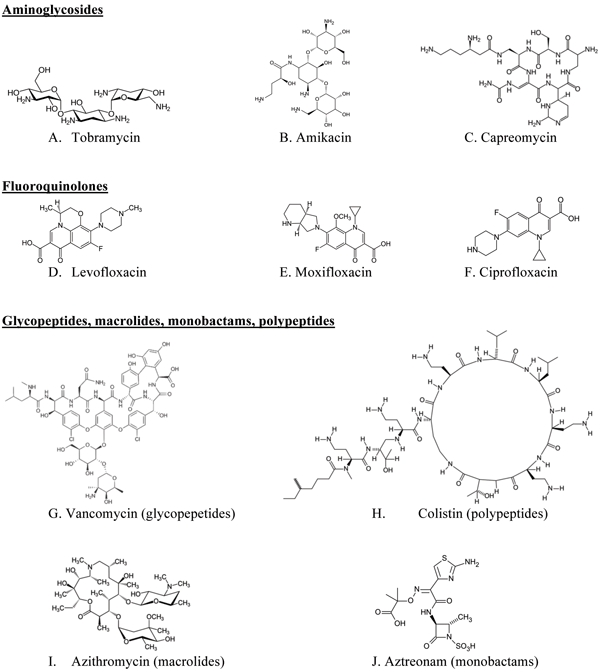
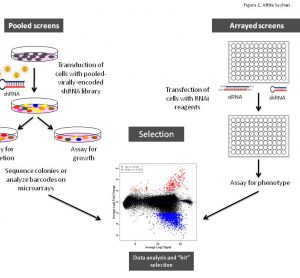
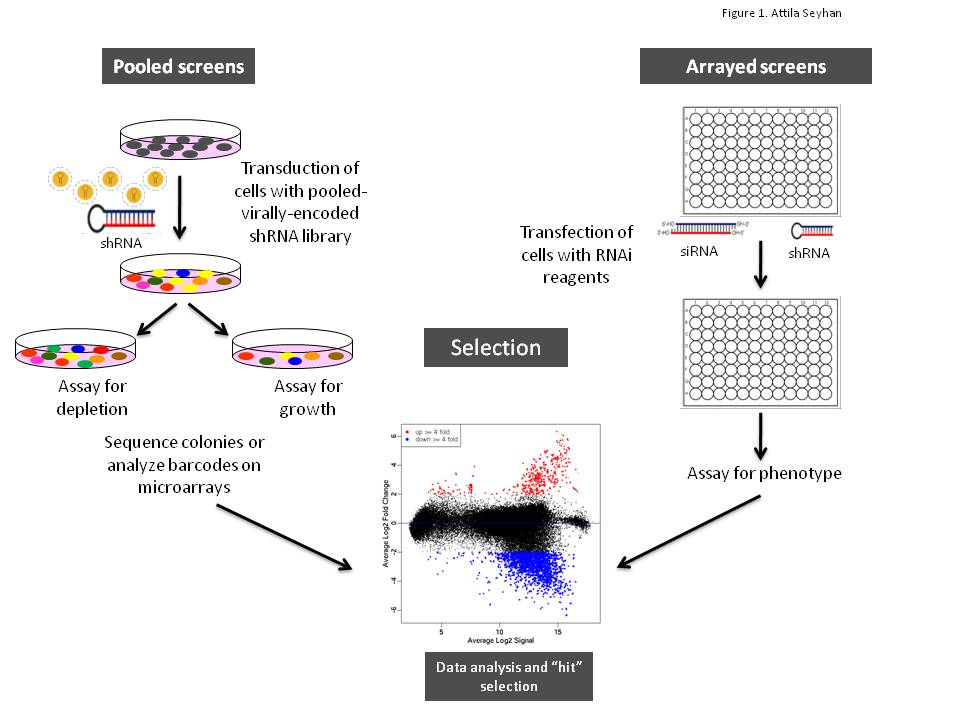
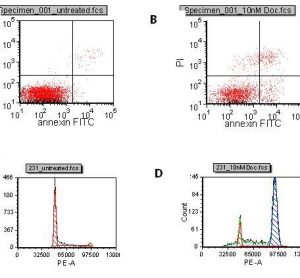
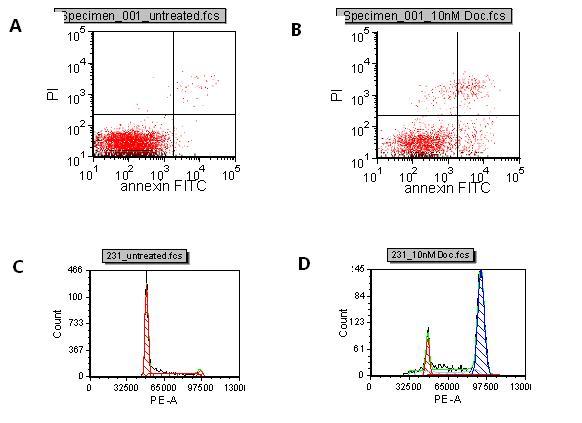
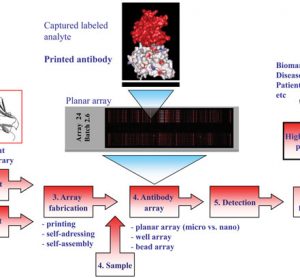
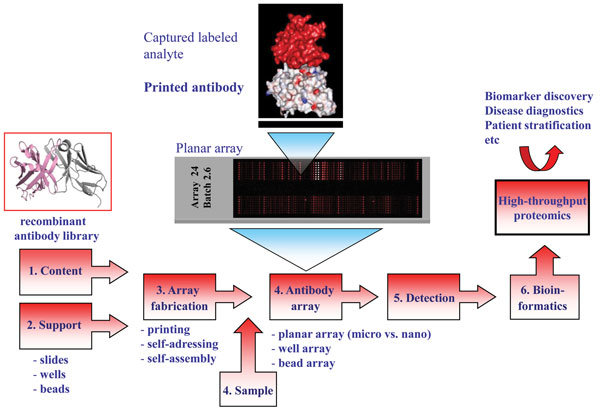
Translational science: The future of medicine
16 February 2011 | By Bahija Jallal, Executive Vice President, Research & Development, MedImmune
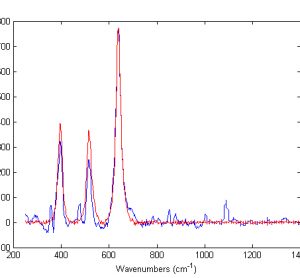
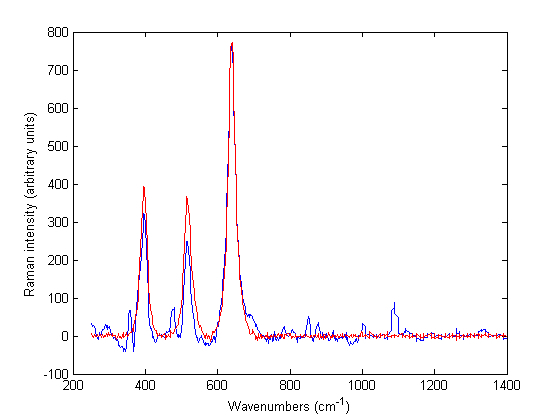
When the term ‘translational medicine’ first came to prominence at the turn of the century, there were suspicions that this concept was simply a rebranding of conventional medical research, describing the process in which scientific discovery at the laboratory bench eventually translates to an effective therapy at the patient’s bedside.…